ℹ️ Skipped - page is already crawled
| Filter | Status | Condition | Details |
|---|---|---|---|
| HTTP status | PASS | download_http_code = 200 | HTTP 200 |
| Age cutoff | PASS | download_stamp > now() - 6 MONTH | 0.3 months ago |
| History drop | PASS | isNull(history_drop_reason) | No drop reason |
| Spam/ban | PASS | fh_dont_index != 1 AND ml_spam_score = 0 | ml_spam_score=0 |
| Canonical | PASS | meta_canonical IS NULL OR = '' OR = src_unparsed | Not set |
| Property | Value | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| URL | https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full | ||||||||||||||||||
| Last Crawled | 2026-04-20 10:24:21 (8 days ago) | ||||||||||||||||||
| First Indexed | 2024-04-05 12:05:18 (2 years ago) | ||||||||||||||||||
| HTTP Status Code | 200 | ||||||||||||||||||
| Content | |||||||||||||||||||
| Meta Title | Frontiers | Psychiatric symptoms in Long-COVID patients: a systematic review | ||||||||||||||||||
| Meta Description | Objective: People who have been infected by COVID-19 showing persistent symptoms after 4 weeks from recovery are thought to suffer from Long-COVID syndrome (..., Objective: People who have been infected by COVID-19 showing persistent symptoms after 4 weeks from recovery are thought to suffer from Long-COVID syndrome (... | ||||||||||||||||||
| Meta Canonical | null | ||||||||||||||||||
| Boilerpipe Text | Abstract
Objective:
People who have been infected by COVID-19 showing persistent symptoms after 4 weeks from recovery are thought to suffer from Long-COVID syndrome (LC). There is uncertainty on the clinical manifestations of LC. We undertook a systematic review to summarize the available evidence about the main psychiatric manifestations of LC.
Method:
PubMed (Medline), Scopus, CINHAL, PsycINFO, and EMBASE were searched until May 2022. Studies reporting estimation of emerging psychiatric symptoms and/or psychiatric diagnoses among adult people with LC were included. Pooled prevalence for each psychiatric condition was calculated in absence of control groups to compare with.
Results:
Thirty-three reports were included in the final selection, corresponding to 282,711 participants with LC. After 4 weeks from COVID-19 infection recovery, participants reported the following psychiatric symptoms: depression, anxiety, post-traumatic symptoms (PTS), cognitive and sleeping disturbances (i.e., insomnia or hypersomnia). The most common psychiatric manifestation resulted to be sleep disturbances, followed by depression, PTS, anxiety, and cognitive impairment (i.e., attention and memory deficits). However, some estimates were affected by important outlier effect played by one study. If study weight was not considered, the most reported condition was anxiety.
Conclusions:
LC may have non-specific psychiatric manifestations. More research is needed to better define LC and to differentiate it from other post-infectious or post-hospitalization syndromes.
Systematic review registration:
PROSPERO (CRD42022299408).
Introduction
Long-COVID syndrome (LC) is a condition that can affect people who have recovered from Coronavirus Disease 2019 (COVID-19). This term was introduced to indicate a set of disorders that persist or occur at from 4 weeks after the elimination of the SARS-CoV-2 virus from the body (
1
). The clinical features of LC are multifaceted; it has been posited that it can affect different organs and systems, causing somatic but also psychological manifestations that impact on quality of life (
2
).
For most people, mild or moderate COVID-19 lasts for about 2 weeks; in some cases, though, symptoms can persist or develop after healing. Furthermore, also in people with asymptomatic infections later health problems may develop (
3
–
5
).
Although progress has been made in the understanding of the clinical and epidemiological features, including the pathogenesis and complications of the acute phase of COVID-19, long-term consequences of the disease remain largely unclear (
6
).
Additionally, while neuropsychiatric symptoms that manifest acutely during infection, such as depression, post-traumatic symptoms [PTS], sleep and cognitive disturbances or anxiety, have received more attention, the medium- and long-term psychiatric outcomes in COVID-19 patients are still little known and understudied (
7
,
8
).
In the available literature, there are highly heterogeneous research works on this topic, applying widely different sample sizes, inclusion and exclusion criteria, and duration of follow-up. In addition, patient assessment is mainly based on various assessment tools and questionnaires, self-administered in most cases, that do not provide a diagnosis of a condition with definite clinical significance.
Therefore, understanding the medium and long-term impact of COVID-19 is still far from being complete, not only in the context of a multidisciplinary approach, but even more so when focusing on specific areas such as mental health (
1
).
We undertook this systematic review to summarize the available evidence about the main psychiatric manifestations of LC. A better understanding of the epidemiology of psychopathological manifestations among LC patients is crucial to develop prevention and early interventions.
Methods
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol of this systematic review was registered with PROSPERO (CRD42022299408).
Data sources and search strategy
We searched the PubMed (Medline), Scopus, CINHAL, PsycINFO, and EMBASE databases until May 2022, using the strategy outlined in the
Supplementary Table 1
of the
Appendix
. In addition, the list of references of the included studies and of other reviews on related topics was screened to identify any other possible study deserving inclusion, and inadvertently missed during the initial literature search. No restrictions regarding language of publication or publication date were set.
Eligibility criteria
We included experimental and observational studies reporting estimation of rates of emerging psychiatric symptoms and/or psychiatric diagnosis among adult people (i.e., ≥18 years old) with LC, without any restriction on other medical comorbidities or setting of enrolment. We excluded studies on participants already suffering from any psychiatric condition, studies assessing the presence of psychiatric symptoms before 4 weeks from COVID-19 recovery, and previous reviews, case-reports, case-series, editorial, and letters to the editor. We only included studies published in peer-reviewed journals, excluding conference abstracts and dissertations. If data from the same sample were published in multiple works, we considered only that study reporting more exhaustive information. Sample overlap was ruled out through a careful check of the registration codes as well as the place and year(s) of sampling.
Where available, outcome data from participants with other inflammatory or infectious diseases, including COVID-19 but without LC, were used as control group.
Terms and definitions
LC was defined as either the presence or the persistence of any symptom that was not present before the infection after 4 weeks from the COVID-19 recovery. Infection from SARS-CoV-2 and recovery from the infection were defined according to the result of the real-time PCR on nasopharyngeal swab sample, or of broncho-alveolar lavage.
Psychiatric symptoms were collected from self-reporting or from validated psychometric tools. Where a psychiatric diagnosis was reported, it had to be defined according to standard operational diagnostic criteria (the Diagnostic and Statistical Manual of Mental Disorders [DSM] or the International Classification of Diseases [ICD]).
Data collection and extraction
Four Reviewers (P.G., V.S., F.R., and F.C.) working independently preliminarily reviewed titles and abstracts of retrieved articles. The initial screening was followed by the analysis of full texts to check compliance with inclusion/exclusion criteria. All disagreements were discussed until consensus, and if consensus was not possible, another member of the team was consulted (M.M.). A standardized form was used for data extraction. Information concerning the year of publication, country, setting, characteristics of study participants (sample size, age, percentages of men and women), LC status, and the presence of psychiatric conditions in the LC groups (and, where available, in the control group) were collected by two authors (P.G. and V.S.) independently. Extraction sheets for each study were cross-checked for consistency and any disagreement was resolved by discussion within the research group.
Statistical analyses
Where possible (i.e., there were at least two studies providing outcome data for LC and controls), quantitative data among studies were summarized using random effects meta-analysis (
9
). To summarize continuous outcome data (i.e., the scores on a psychometric tool), the pooled Hedges' g standardized mean differences (SMDs) and the corresponding 95% confidence intervals (CIs) were applied, while pooled odds ratios (ORs) and the corresponding 95% CIs (
10
) were used to report on dichotomous outcome data (i.e., presence/absence of psychiatric diagnosis or psychiatric symptoms).
If meta-analysis was not possible, we calculated the pooled prevalence of psychiatric symptoms and/or psychiatric diagnosis among LC patients. These estimates consisted in weighted-mean prevalence, raw mean prevalence, and median prevalence, with the relative lower and upper ranges across the studies included in the final selection.
The analyses were performed in R (
11
). Statistical tests were 2-sided and used a significance threshold of
p
< 0.05.
Risk of bias assessment and the GRADE
Bias risk in the included studies was independently assessed by three reviewers (P.G., V.S., and F.R.), using the Cochrane risk of bias tool (
12
). All disagreements were discussed until consensus, and if necessary, another member of the team was consulted (M.M.). Each item on the risk of bias assessment was scored as high, low, or unclear, and the GRADE tool was used to assess the overall certainty of evidence (
13
). Further information is available in the
Supplementary material
.
Results
Study characteristics
As shown in
Figure 1
, from 2078 records screened on title and abstract, 114 full texts were analyzed. The review process led to the selection of 33 studies (
3
,
4
,
6
–
8
,
14
–
41
). These studies, referring to 33 different samples and involving a total of 282,711 LC participants, were included in the final selection and quantitative synthesis.
Figure 1
On an average, across the studies, 48% of participants were females (range 23.1–100%). The mean age of participants was 53.2 years (range 33.7–73.2). The selected studies were conducted in 13 countries: US (
n
= 6; 18.2%); Italy (
n
= 5; 15.2%); Egypt, France, Netherlands, Spain, UK (each
n
= 3; 9.1%); India (
n
= 2; 6.1%); Austria, China, Germany, Iran, Mexico (each
n
= 1; 3.0%).
All the studies were published in the last 2 years: 2021 (
n
= 31; 93.9%); 2022 (
n
= 2; 6.1%).
With respect to the outcomes reported, only 2 studies (6.1%) provided data about psychiatric diagnosis: one study assessed depressive and anxiety disorder (GAD) through a clinical interview, the other study used retrospective screening of the electronic clinical records to investigate prevalence of anxiety and depression, and cognitive impairment, according to the ICD-10 system. The remaining studies (
n
= 31, 93.9%) used self-reporting or other psychometric tools to measure the level of: depression or anxiety (
n
= 26, 78.8%), cognitive impairment (
n
= 16, 48.5%), PTS (
n
= 13, 39.4%), and sleep disturbances (
n
= 18, 54.5%). These studies applied dichotomization into positive/negative at the psychometric assessment based on the scales' cut-off for clinical significance, and the estimated prevalence for each study was calculated as the number of participants with score above the cut-off divided to the total number of participants assessed.
Notably, none of the studies included in the final selection applied a control group without LC. Concerning the severity of COVID-19 infection, 15 studies (45.5%) included patients hospitalized due to COVID-19 infection, 5 studies (15.2%) were performed on patients who had mild infection not requiring hospitalization, and 11 studies (33.3%) included both hospitalized and other managed outpatients. Information about infection severity was missing in 2 studies (6.1%).
All studies characteristics are summarized in
Table 1
.
Table 1
Author, year
Country
Study design
Females %
Mean age
N
Severity of COVID-19 infection
P depression (measure)
P Anxiety (measure)
P Cognitive impairment (measure)
P PTS (measure)
P Sleep disturbances (measure)
Ahmed et al. (
14
)
Egypt
Cohort
54%
46.5
182
Both hospitalized and outpatients
0.374 (SCL-90)
0.619 (SCL-90)
NR
0.286 (PCL-5)
0.648 (PSQI)
Aly and Saber (
15
)
Egypt
Cross-sectional
100%
73.2
115
NR
NR
NR
0.252 (self-reported)
NR
0.243 (self-reported)
Aranda et al. (
16
)
Spain
Cohort
30%
64
113
Hospitalized
0.301 (BDI)
0.487 (STAI)
NR
0.788 (IES-R)
0.292 (NR)
Bai et al. (
17
)
Italy
Cohort
36%
57
377
Both hospitalized and outpatients
0.106 (HADS)
0.188 (HADS)
0.202 (NR)
0.225 (IES-R)
NR
Boesl et al. (
18
)
Italy
Cohort
67%
45.8
100
Mild
0.615 (BDI)
NR
0.306 (MoCA)
NR
0.337 (ESS)
De Graaf et al. (
3
)
Netherlands
Cohort
37%
60.8
81
Both hospitalized and outpatients
0.123 (PHQ-9)
0.037 (GAD-7)
0.160 (CFQ-25)
0.062 (PCL-5)
NR
Evans et al. (
4
)
UK
Cohort
36%
57.9
1077
Hospitalized
0.262 (PHQ-9)
0.235 (GAD-7)
0.139 (NR)
0.117 (PCL-5)
NR
Frontera et al. (
19
)
US
Case-control
35%
68.5
280
Hospitalized
0.254 (NeuroQoL)
0.511 (NeuroQoL)
0.473 (MoCA)
NR
0.375 (NeuroQoL)
Ganesh et al. (
20
)
US
Cohort
61%
44
817
Both hospitalized and outpatients
NR
NR
NR
NR
0.200 (PROMIS)
Garjani et al. (
21
)
UK
Cohort
82%
50
165
Mild
0.504 (NR)
†
NR
NR
NR
NR
Gonzàlez-Hermosillo et al. (
22
)
Mexico
Cohort
35%
51
130
Hospitalized
0.354 (self-reported)
0.392 (self-reported)
0.454 (self-reported)
NR
0.454 (self-reported)
Gouraud et al. (
23
)
France
Cohort
29%
60
100
Hospitalized
0.220 (HADS)
0.310 (HADS)
NR
NR
NR
Graham et al. (
24
)
US
Case-control
66%
43.7
50
Mild
0.400 (NR)
†
NR
0.820 (PROMIS)
NR
0.360 (PROMIS)
Gramaglia et al. (
6
)
Italy
Cohort
40%
61
238
Both hospitalized and outpatients
0.294 (MINI)
0.328 (MINI)
NR
0.429 (NR)
NR
Horwitz et al. (
7
)
US
Cohort
40%
62
126
Hospitalized
NR
NR
0.413 (PROMIS)
NR
0.349 (PROMIS)
Huang et al. (
8
)
China
Cohort
48%
57
1733
Hospitalized
0.227 (interview)
†
NR
NR
NR
0.264 (interview)
Imran et al. (
25
)
India
Cross-sectional
33%
44.5
103
Hospitalized
0.126 (PHQ-9)
0.214 (GAD-7)
NR
0.087 (PCL-5)
NR
Lemhofer et al. (
26
)
Germany
Cross-sectional
59%
49.8
365
Mild
NR
0.249 (NR)
NR
NR
0.301 (NR)
Lombardo et al. (
27
)
Italy
Cohort
54%
53
303
Both hospitalized and outpatients
NR
NR
0.363 (semi-structured interview)
NR
0.465 (semi-structured interview)
Mendez et al. (
28
)
Spain
Cohort
41%
57
179
Hospitalized
0.268 (PHQ-9)
0.296 (GAD-7)
0.184 (NR)
0.251 (DTS)
NR
Morin et al. (
37
)
France
Cohort
42%
60.9
478
Hospitalized
0.206 (BDI)
0.314 (HADS)
0.384 (MoCA, D2-R, Q3PC)
0.142 (PCL-5)
0.536 (ISI)
Naik et al. (
29
)
India
Cohort
31%
41.6
272
Both hospitalized and outpatients
0.022 (interview)
0.029 (interview)
NR
NR
0.063 (interview)
Rass et al. (
30
)
Austria
Cohort
39%
55
90
Both hospitalized and outpatients
0.121 (HADS)
0.222 (HADS)
NR
0.100 (PCL-5)
NR
Romero-Duarte et al. (
31
)
Spain
Cohort
46%
63
794
Hospitalized
0.044 (NR)
NR
NR
NR
0.049 (NR)
Scherlinger et al. (
32
)
France
Case-control
66%
40
30
Mild
0.100 (Psychological interview)
0.267 (Psychological interview)
NR
0.300 (PCL-5)
NR
Simani et al. (
33
)
Iran
Cohort
33%
54.62
120
Hospitalized
NR
NR
NR
0.058 (PCL-5)
NR
Sykes et al., (
34
)
UK
Cohort
34%
134
Hospitalized
0.396 (self-reported)
0.478 (self-reported)
0.097 (self-reported)
NR
0.351 (self-reported)
Taquet et al. (
35
)
Netherlands
Cohort
58%
39.4
273618
Both hospitalized and outpatients
0.155 (ICD-10)
†
NR
0.040 (ICD-10)
NR
NR
Tawfik (
36
)
Egypt
Cohort
58%
33.7
120
Both hospitalized and outpatients
NR
NR
0.008 (NR)
NR
0.042 (NR)
Van den Borst et al. (
38
)
Netherlands
Cohort
40%
59
124
Both hospitalized and outpatients
0.117 (HADS)
0.100 (HADS)
NR
0.073 (PCL-5)
NR
Vanichkachorn et al. (
39
)
US
Cohort
68%
45.7
100
NR
NR
NR
0.005 (NR)
NR
0.003 (NR)
Vannorsdall et al. (
40
)
US
Cohort
59%
54.5
82
Hospitalized
0.902 (PHQ-9)
0.646 (GAD-7)
0.805 (QDRS)
0.25 (IES-R)
NR
Vassalini et al. (
41
)
Italy
Cohort
46%
57
115
Hospitalized
0.148 (PHQ-9)
NR
NR
NR
NR
Characteristics of the included studies.
P, prevalence; PTS, post-traumatic symptoms; NR, not-reported; SCL-90, symptom checklist 90; PCL-5, PTSD checklist for DSM-5; PSQI, Pittsburgh sleep quality index; BDI, Beck depression inventory; STAI, state-trait anxiety inventory; IES-R, impact of event scale—revised; HADS, hospital anxiety and depression scale; MoCA, Montreal cognitive assessment; ESS, Epworth sleepiness scale; PHQ-9, patient health questionnaire-9; GAD-7, general anxiety disorder-7; CFQ-25, cognitive failure questionnaire-25; UK, United Kingdom; US, United States of America; NeuroQoL, health-related quality of life for clinical research in neurology; PROMIS, patient-reported outcomes measurement information system; MINI, mini-international neuropsychiatric interview; DTS, Davidson trauma scale; D2-R, D2 test of attention—revised; Q3PC, Q3PC cognitive screening questionnaire; ISI, insomnia severity index; ICD-10, international classification of diseases 10th revision; QDRS, quick dementia rating scale.
†
depression and anxiety aggregated prevalence.
Prevalence of psychiatric symptoms across the studies
Table 2
summarize the pooled prevalence estimates for each psychiatric symptom across the studies included in this review, and the population prevalence worldwide. Notably, prevalence of depression, anxiety, cognitive impairment, PTS, and sleep disturbances resulted much higher among LC patients than in the general population (
42
–
46
).
Table 2
Outcome
Weighted mean P
Mean P (min; max)
Median P
N studies (LC participants)
World P
Depression
0.212
0.254 (0.022; 0.902)
0.220
21 (5,079)
0.038
Anxiety
0.158
0.313 (0.029; 0.646)
0.296
23 (28,001)
0.040
Cognitive impairment
0.042
0.269 (0.005; 0.820)
0.227
16 (277,268)
0.011
PTS
0.192
0.218 (0.058; 0.788)
0.130
13 (3,162)
0.036
Sleeping disturbances
0.270
0.296 (0.003; 0.648)
0.319
18 (6,212)
0.038
Pooled prevalence of psychiatric symptoms across the included studies and worldwide prevalence.
P, prevalence; min, minimum; max, maximum; LC, Long-COVID syndrome; NA, not available; PTS, post-traumatic symptoms.
Prevalence of depression
Twenty-one studies (63.3%) provided outcome data for depression among LC patients. The weighted mean prevalence across the studies was 0.212, that is quite similar to the unweighted mean and median prevalence (0.254 [range: 0.022–0.902] and 0.220, respectively), consistent with not significant outlier effect played by any of the study in the pooled estimate.
Figure 2
shows comparison of the depression prevalence estimates across the studies, and the weighted mean prevalence.
Figure 2
Prevalence of anxiety
Twenty-three studies (69.7%) provided outcome data for anxiety among LC patients. The weighted mean prevalence across the studies was 0.158 and was markedly influenced by the study from Taquet et al. (
35
) with a far larger sample size. Unweighted mean and median prevalence were 0.313 (range: 0.029–0.646) and 0.296, respectively.
Figure 3
shows comparison of the anxiety prevalence estimates across the studies, and the weighted mean prevalence.
Figure 3
Prevalence of cognitive impairment
Sixteen studies (48.5%) provided outcome data for cognitive impairment among LC patients. The weighted mean prevalence across the studies was 0.042 and, again, was markedly influenced by the study from Taquet et al. (
35
) with the largest sample size and providing among the three lowest estimates of anxiety prevalence. Unweighted mean and median prevalence were 0.269 (range: 0.005–0.820) and 0.227, respectively.
Figure 4
shows comparison of the cognitive impairment prevalence estimates across the studies, and the weighted mean prevalence.
Figure 4
Prevalence of PTS
Thirteen studies (39.4%) provided outcome data for PTS among LC patients. The weighted mean prevalence across the studies was 0.192. Unweighted mean and median prevalence were 0.218 (range: 0.058–0.788) and 0.130, respectively.
Figure 5
shows comparison of the PTS prevalence estimates across the studies, and the weighted mean prevalence.
Figure 5
Prevalence of sleep disturbances
Eighteen studies (54.5%) provided outcome data for sleep disturbances among LC patients. The weighted mean prevalence across the studies was 0.270. Unweighted mean and median prevalence were 0.296 (range: 0.003–0.648) and 0.319, respectively.
Figure 6
shows comparison of the sleep disturbances prevalence estimates across the studies, and the weighted mean prevalence.
Figure 6
Psychiatric symptoms by COVID-19 infection severity
We examined the potential association between the severity of COVID-19 infection and the occurrence of psychiatric symptoms. For this purpose, we considered hospitalization as a proxy for severe infection, and outpatient management as indicator of mild infection. As shown in
Table 3
, the comparison of the severe, mild, and both mild and severe groups in terms of average prevalence of psychiatric symptoms did not find any statistically significant difference, suggesting that the severity of the infection is not related to the development of later psychiatric symptoms. Further analysis was conducted to compare only mild and severe patients, with the severe group consisting of studies that included at least one hospitalized patient. This analysis confirmed that there were no statistically significant differences in depression, anxiety, PTS, and sleep disturbances, but found inverse association between the severity of the infection and cognitive complaints (
p
= 0.048).
Table 3
Mean P (SD) mild
Mean P (SD) severe
Mean P (SD) both
p
-value
N
5
15
11
Depression
0.36 (0.36)
0.29 (0.22)
0.16 (0.12)
0.339
Anxiety
0.36 (0.12)
0.37 (0.14)
0.21 (0.19)
0.089
Cognitive impairment
0.56 (0.36)
0.31 (0.16)
0.15 (0.14)
0.061
PTS
NA
0.24 (0.28)
0.20 (0.15)
0.731
Sleeping disturbances
0.33 (0.03)
0.33 (0.14)
0.28 (0.26)
0.879
Prevalence of psychiatric symptoms by severity of the COVID-19 infection.
P, prevalence; SD, standard deviation; N, number; PTS, post-traumatic symptoms; NA, not available.
p-value are based on multiple groups analysis of variance (ANOVA).
Risk of bias and GRADE
A detailed summary on the risk of bias in all 33 trials has been reported in the
Appendix
(see
Supplementary Figures 1
,
2
), along with an assessment of the quality of the evidence (see
Supplementary Table 2
). In the GRADE system, the evidence from observational studies is initially set to low, there are then criteria that can be used either to downgrade or upgrade (see further information in the
Appendix
). The quality of the evidence is rated very low with serious threats related to the risk of bias and inconsistency.
Discussion
This study set out to investigate the prevalence of psychiatric symptoms among LC patients. We found that the symptoms mostly associated with LC were depression, anxiety, cognitive and sleep disturbances, and PTS. The prevalence of these symptoms among LC patients is remarkably higher than that in the general population.
However, it is necessary to underline that, among the studies included in the final selection, there was one study (
35
) that had a sample size accounting for around 96% of the total number of participants included in this review. That study provided estimates for anxiety and cognitive impairments and the weighted average for these outcomes falls exactly on the value of the prevalence estimated by Taquet et al. (
35
) by looking at the forest plots of anxiety and cognitive impairment, it can be easily observed that only three studies for anxiety and two studies for cognitive impairment provided estimates smaller than Taquet et al. (
35
) supporting its influence on the pooled prevalence. Accordingly, for these two outcomes, the raw mean resulted higher than the weighted mean, because the former is not affected by the differences in the sample size across the studies.
Nevertheless, the relatively high pooled prevalence of psychiatric symptoms among LC patients requires a better understanding. Our analyses did not find a significant association between the severity of COVID-19 infection and psychiatric symptoms, except for a potential inverse association with symptoms of cognitive impairment. These findings align with the conclusions drawn from most of the reports included in our systematic review, which examined the relationship between infection severity and psychiatric symptoms (
6
,
17
,
20
,
23
,
27
,
34
,
40
). Notably, only one study reported an increased risk of depression and anxiety among individuals with the most severe form of infection (
8
). However, it is important to acknowledge that the confidence in the results is limited by the comparatively small representation of patients with mild infection, leading to low statistical power.
It should be noted that research in the pre-COVID-19 era observed that survivors after intensive care (IC) are at greater risk of developing long-term mental disorders (
47
). Particularly, anxiety, depression, and PTSD would have occurred in half of this sample of UK patients discharged from IC. Psychiatric symptoms of the post-IC syndrome fall into three broad categories: physical, cognitive, and psychological deterioration. Symptoms of physical deterioration include fatigue and insomnia, while cognitive and psychiatric symptoms include anxiety, depression, memory impairment, and PTSD. Therefore, there seems to be a significant overlap in the experience of some of the LC patients analyzed in this review with post-IC syndrome.
A considerable amount of COVID-related research also focused on the effect on mental health of public health measures (such as quarantine, lock-down, social isolation and other limitations to personal freedom), finding an association with symptoms of depression, anxiety, loneliness, psychosocial distress, and persisting post-traumatic arousal (
48
,
49
). Therefore, another possible explanation for the higher prevalence of psychiatric symptoms among LC patients may be more a consequence of the imposed quarantine and other restrictions in terms of anxiety, fear, anger, and other negative emotions, regardless of specific aspects of the COVID-19 infection such as neuro- or systemic inflammation. Even if the quarantine imposed by a local health authority has not been directly associated with any psychological outcomes (
50
), it was suggested that belonging to a publicly recognized COVID-19 risk group/community would be associated with increased anxiety, depressive symptoms, self-concern, fear, increased psychosocial distress, and decreased life satisfaction. In addition, loneliness and isolation have been associated with an increased risk for various mental disorders (as well as for various somatic diseases). In the context of the COVID-19 pandemic, loneliness has been found to be predictive of depressive and anxious symptoms during the lockdown measures (
51
,
52
).
COVID-19 is in fact only the most recent of many other infectious diseases that have been associated with chronic sequelae after recovering from the acute phase of infection (
53
): similarly as with LC, the underlying pathophysiological and etiological mechanisms are far from being clearly understood. The review by Choutka et al. (
53
) investigated the common characteristics between LC and other chronic infectious syndromes, finding higher prevalence of the following symptoms: intolerance to physical effort, neurocognitive and sensory impairment, persistent flu-like symptoms, disturbed sleep, myalgias, and arthralgias. The greatest analogies are with the post-acute effects described in the SARS epidemic in 2002–2004 (
54
).
All these elements are probably interrelated with each other and influential in the experience of LC patients.
Limitations
The results of this review should be interpreted considering its limitations. First, the lack of a control group made difficult to draw considerations on the risk of psychiatric symptoms among LC patients, reducing considerably generalizability and reliability of our findings. This translated also in the impossibility to meta-analyze the results of the selected studies to detail the risk of psychiatric symptoms in patients who have had COVID-19. Second, most of the included studies reported measures of psychiatric symptoms instead of assessing psychiatric diagnoses, with a risk for diagnostic overestimation: this was partially attenuated by including only studies applying validated psychometric tools or clinical interviews. Third, there were a marked outlier effect played by one study (
35
), which implemented a sample size accounting for more than 90% of the total sample size. Even though that study resulted at low risk of bias in the assessment, it may have impacted on the pooled prevalence estimate of the outcomes reported in that study. Fourth, the condition of LC has been assessed only through a temporal criterion, that was 4 weeks after recovery from the infection. The lack of a more comprehensive definition of LC may have increase the heterogeneity in the estimates. Finally, the risk of bias was rated high or unclear in many studies, with serious threats related to the inconsistency in the estimates and to the assessment of confounders.
Implications for research and clinical practice
The overlapping of some clinical features of LC in terms of signs and symptoms with other post-infectious syndromes and with the post-IC syndrome would suggest the involvement of shared pathophysiological pathways. The perspective of identifying a unified etiological model would lead the way toward the implementation of diagnostic markers and tailored treatments (
53
). At present, however, our understanding of the underlying pathophysiological mechanisms and etiological factors is poor, though promising studies are being conducted (
55
–
57
). For example, a recent review advanced the hypothesis that perivascular inflammation serves as the critical pathogenetic factor for LC neuropsychiatric manifestations (
58
). Indeed, SARS-CoV-2 and other viruses (such as retrovirus) showed the ability to activate brain mast cells and microglia resulting in the release of inflammatory, neurotoxic, and vasoactive mediators impacting neuronal connectivity and signal transmission (
59
–
61
). Hopefully, that may also converge to a better definition of functional and psycho-somatic syndromes, such as fibromyalgia and chronic fatigue syndrome, for which the association with viral infections has been previously proposed (
62
–
67
).
More research is therefore needed, more clearly comparing different patient groups (e.g., LC patients that were admitted to ICU vs. other ICU patients with or without other infectious diseases; LC patients vs. patients remitting from other infectious diseases) and applying prospective designs, allowing causal considerations, and providing more epidemiological details. Also, qualitative studies investigating the subjective experience of people recovered from COVID-19 are being conducted (
68
): this approach may also contribute to the understanding of the psychological mechanism contributing to the onset of psychological symptoms. Such different research methods could converge on a better conceptualization and analysis of the symptoms associated with the LC syndrome, as well as supporting the construction of a better defined and unified nomenclature.
A better understanding of the LC psycho-pathophysiology is essential to provide and improve treatment. From a therapeutic point of view, in close relation both to the traumatic component of a part of the symptoms found in LC, and to the inflammatory component (initially exerted by the infection and then self-sustained), interventions aimed at reducing the inflammatory process and reducing the excessive activation of the sympathetic nervous system through a relaxation response may be useful. For example, models of intervention involving reconditioning and mindfulness may help patients suffering from LC (
69
). Future clinical trials on LC patients may be therefore warranted.
Conclusions
People who have recovered from COVID-19 may experience more and persistent psychiatric symptoms. These include depression, anxiety, post-traumatic distress, cognitive and sleeping disturbances. However, there is marked heterogeneity in the literature about how these symptoms are investigated and differentiated from other post-infectious or post-hospitalization conditions. More research, particularly implementing control groups and prospective follow-up, are needed to better define psychopathology related or included into the LC syndrome.
Statements
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
MM, PG, and VS: conceptualization and planning and interpretation of the results. PG, VS, FC, FR, and PM: acquisition. MM: analysis of the data. MM, PG, VS, FC, FR, and PM: drafting. SF, LP, and GG: critical revision of the manuscript. All authors approved the final submitted version of the manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at:
https://www.frontiersin.org/articles/10.3389/fpsyt.2023.1138389/full#supplementary-material
References
1.
Akbarialiabad
H
Taghrir
MH
Abdollahi
A
Ghahramani
N
Kumar
M
Paydar
S
et al
. Long COVID, a comprehensive systematic scoping review.
Infection.
(2021) 49:1163–86. 10.1007/s15010-021-01666-x
Pubmed Abstract
CrossRef
Google Scholar
2.
Michelen
M
Manoharan
L
Elkheir
N
Cheng
V
Dagens
A
Hastie
C
et al
. Characterising long COVID: a living systematic review.
BMJ Glob Health.
(2021) 6:e005427. 10.1136/bmjgh-2021-005427
Pubmed Abstract
CrossRef
Google Scholar
3.
De Graaf
MA
Antoni
ML
Kuile
MM
Arbous
MS
Duinisveld
AJF
Feltkamp
MCW
et al
. Short-term outpatient follow-up of COVID-19 patients: a multidisciplinary approach.
eClinicalMedicine.
(2021) 32:25. 10.1016/j.eclinm.2021.100731
Pubmed Abstract
CrossRef
Google Scholar
4.
Evans
RA
McAuley
H
Harrison
EM
Shikotra
A
Singapuri
A
Sereno
M
et al
. Physical, cognitive, and mental health impacts of COVID-19 after hospitalisation (PHOSP-COVID): a UK multicentre, prospective cohort study.
Lancet Respir Med.
(2021) 9:1275–87. 10.1016/S2213-2600(21)00383-0
Pubmed Abstract
CrossRef
Google Scholar
5.
Fiore
G
Ferrari
S
Cutino
A
Giorgino
C
Valeo
L
Galeazzi
GM
et al
. Delirium in COVID-19 and post-liver transplant patients: an observational study.
Int J Psychiatry Clin Pract.
(2022) 26:343–51. 10.1080/13651501.2022.2026403
Pubmed Abstract
CrossRef
Google Scholar
6.
Gramaglia
C
Gambaro
E
Bellan
M
Balbo
PE
Baricich
A
Sainaghi
PP
et al
. Mid-term psychiatric outcomes of patients recovered from COVID-19 from an italian cohort of hospitalized patients.
Front Psychiatry.
(2021) 12:667385. 10.3389/fpsyt.2021.667385
Pubmed Abstract
CrossRef
Google Scholar
7.
Horwitz
LI
Garry
K
Prete
AM
Sharma
S
Mendoza
F
Kahan
T
et al
. Six-month outcomes in patients hospitalized with severe COVID-19.
J Gen Intern Med.
(2021) 36:3772–7. 10.1007/s11606-021-07032-9
Pubmed Abstract
CrossRef
Google Scholar
8.
Huang
C
Huang
L
Wang
Y
Li
X
Ren
L
Gu
X
et al
. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study.
Lancet.
(2021) 397:220–32. 10.1016/S0140-6736(20)32656-8
Pubmed Abstract
CrossRef
Google Scholar
9.
DerSimonian
R
Laird
N
. Meta-analysis in clinical trials.
Control Clin Trials.
(1986) 7:177–88. 10.1016/0197-2456(86)90046-2
Pubmed Abstract
CrossRef
Google Scholar
10.
Higgins
J
Thomas
J
Chandler
J
Cumpston
M
Li
T
Page
M
.
Cochrane Handbook for Systematic Reviews of Interventions. version 6.2. Cochrane
(2021). Available online at:
www.training.cochrane.org/handbook
. (accessed January 28, 2022).
Google Scholar
11.
RStudio Team
.
RStudio: Integrated Development Environment for R.
(2021) Available online at:
http://www.rstudio.com/
Google Scholar
12.
Higgins
JPT
Altman
DG
Gøtzsche
PC
Jüni
P
Moher
D
Oxman
AD
et al
. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials.
BMJ.
(2011) 343:d5928. 10.1136/bmj.d5928
Pubmed Abstract
CrossRef
Google Scholar
13.
Schünemann
H
Brozek
J
Guyatt
G
Oxman
A
.
GRADE Handbook for Grading Quality of Evidence Strength of Recommendations.
(2013). Available online at:
https://gdt.gradepro.org/app/handbook/handbook.html
(accessed July 6, 2022).
Google Scholar
14.
Ahmed
GK
Khedr
EM
Hamad
DA
Meshref
TS
Hashem
MM
Aly
MM
. Long term impact of Covid-19 infection on sleep and mental health: a cross-sectional study.
Psychiatry Res.
(2021) 305:114243. 10.1016/j.psychres.2021.114243
Pubmed Abstract
CrossRef
Google Scholar
15.
Aly
MAEG
Saber
HG
. Long COVID and chronic fatigue syndrome: a survey of elderly female survivors in Egypt.
Int J Clin Pract.
(2021) 75:e14886. 10.1111/ijcp.14886
Pubmed Abstract
CrossRef
Google Scholar
16.
Aranda
J
Oriol
I
Martín
M
Feria
L
Vázquez
N
Rhyman
N
et al
. Long-term impact of COVID-19 associated acute respiratory distress syndrome.
J Infect.
(2021) 83:581–8. 10.1016/j.jinf.2021.08.018
Pubmed Abstract
CrossRef
Google Scholar
17.
Bai
F
Tomasoni
D
Falcinella
C
Barbanotti
D
Castoldi
R
Mulè
G
et al
. Female gender is associated with long COVID syndrome: a prospective cohort study.
Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis.
(2022) 28:611. 10.1016/j.cmi.2021.11.002
Pubmed Abstract
CrossRef
Google Scholar
18.
Boesl
F
Audebert
H
Endres
M
Prüss
H
Franke
C
. A neurological outpatient clinic for patients with post-COVID-19 syndrome—a report on the clinical presentations of the first 100 patients.
Front Neurol.
(2021) 12:405. 10.3389/fneur.2021.738405
Pubmed Abstract
CrossRef
Google Scholar
19.
Frontera
JA
Yang
D
Lewis
A
Patel
P
Medicherla
C
Arena
V
et al
. A prospective study of long-term outcomes among hospitalized COVID-19 patients with and without neurological complications.
J Neurol Sci.
(2021) 426:7486. 10.1016/j.jns.2021.117486
Pubmed Abstract
CrossRef
Google Scholar
20.
Ganesh
R
Ghosh
AK
Nyman
MA
Croghan
IT
Grach
SL
Anstine
CV
et al
. Scales for assessment of persistent post-COVID symptoms: a cross sectional study.
J Prim Care Community Health.
(2021) 12:21501327211030412. 10.1177/21501327211030413
Pubmed Abstract
CrossRef
Google Scholar
21.
Garjani
A
Middleton
RM
Nicholas
R
Evangelou
N
. Recovery from COVID-19 in multiple sclerosis: a prospective and longitudinal cohort study of the united kingdom multiple sclerosis register.
Neurol—Neuroimmunol Neuroinflamm.
(2022) 9:1118. 10.1212/NXI.0000000000001118
Pubmed Abstract
CrossRef
Google Scholar
22.
González-Hermosillo
JA
Martínez-López
JP
Carrillo-Lampón
SA
Ruiz-Ojeda
D
Herrera-Ramírez
S
Amezcua-Guerra
LM
et al
. del R Post-Acute COVID-19 Symptoms, a Potential Link with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A 6-Month Survey in a Mexican.
Cohort Brain Sci.
(2021) 11:760. 10.3390/brainsci11060760
Pubmed Abstract
CrossRef
Google Scholar
23.
Gouraud
C
Bottemanne
H
Lahlou-Laforêt
K
Blanchard
A
Günther
S
Batti
SE
et al
. association between psychological distress, cognitive complaints, and neuropsychological status after a severe COVID-19 episode: a cross-sectional study.
Front Psychiatr.
(2021) 12:861. 10.3389/fpsyt.2021.725861
Pubmed Abstract
CrossRef
Google Scholar
24.
Graham
EL
Clark
JR
Orban
ZS
Lim
PH
Szymanski
AL
Taylor
C
et al
. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized COVID-19 “long haulers”.
Ann Clin Transl Neurol.
(2021) 8:1073–85. 10.1002/acn3.51350
Pubmed Abstract
CrossRef
Google Scholar
25.
Imran
J
Nasa
P
Alexander
L
Upadhyay
S
Alanduru
V
. Psychological distress among survivors of moderate-to-critical COVID-19 illness: a multicentric prospective cross-sectional study.
Indian J Psychiatry.
(2021) 63:285–9. 10.4103/psychiatry.IndianJPsychiatry_1074_20
Pubmed Abstract
CrossRef
Google Scholar
26.
Lemhöfer
C
Sturm
C
Loudovici-Krug
D
Best
N
Gutenbrunner
C
. The impact of Post-COVID-syndrome on functioning—results from a community survey in patients after mild and moderate SARS-CoV-2-infections in Germany.
J Occup Med Toxicol.
(2021) 16:45. 10.1186/s12995-021-00337-9
Pubmed Abstract
CrossRef
Google Scholar
27.
Lombardo
MDM
Foppiani
A
Peretti
GM
Mangiavini
L
Battezzati
A
Bertoli
S
et al
. Long-Term coronavirus disease 2019 complications in inpatients and outpatients: a one-year follow-up cohort study.
Open Forum Infect Dis.
(2021) 8:ofab384. 10.1093/ofid/ofab384
Pubmed Abstract
CrossRef
Google Scholar
28.
Méndez
R
Balanzá-Martínez
V
Luperdi
SC
Estrada
I
Latorre
A
González-Jiménez
P
et al
. Short-term neuropsychiatric outcomes and quality of life in COVID-19 survivors.
J Intern Med.
(2021) 290:621–31. 10.1111/joim.13262
Pubmed Abstract
CrossRef
Google Scholar
29.
Naik
S
Haldar
SN
Soneja
M
Mundadan
NG
Garg
P
Mittal
A
et al
. Post COVID-19 sequelae: a prospective observational study from Northern India.
Drug Discov Ther.
(2021) 15:254–60. 10.5582/ddt.2021.01093
Pubmed Abstract
CrossRef
Google Scholar
30.
Rass
V
Ianosi
B-A
Zamarian
L
Beer
R
Sahanic
S
Lindner
A
et al
. Factors associated with impaired quality of life three months after being diagnosed with COVID-19.
Qual Life Res.
(2022) 31:1401–14. 10.1007/s11136-021-02998-9
Pubmed Abstract
CrossRef
Google Scholar
31.
Romero-Duarte
Á
Rivera-Izquierdo
M
Guerrero-Fernández de Alba
I
Pérez-Contreras
M
Fernández-Martínez
NF
Ruiz-Montero
R
et al
. Sequelae, persistent symptomatology and outcomes after COVID-19 hospitalization: the ANCOHVID multicentre 6-month follow-up study.
BMC Med.
(2021) 19:129. 10.1186/s12916-021-02003-7
Pubmed Abstract
CrossRef
Google Scholar
32.
Scherlinger
M
Felten
R
Gallais
F
Nazon
C
Chatelus
E
Pijnenburg
L
et al
. Refining “Long-COVID” by a prospective multimodal evaluation of patients with long-term symptoms attributed to SARS-CoV-2 infection.
Infect Dis Ther.
(2021) 10:1747–63. 10.1007/s40121-021-00484-w
Pubmed Abstract
CrossRef
Google Scholar
33.
Simani
L
Ramezani
M
Darazam
IA
Sagharichi
M
Aalipour
MA
Ghorbani
F
et al
. Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19.
J Neurovirol.
(2021) 27:154–9. 10.1007/s13365-021-00949-1
Pubmed Abstract
CrossRef
Google Scholar
34.
Sykes
DL
Holdsworth
L
Jawad
N
Gunasekera
P
Morice
AH
Crooks
MG
. Post-COVID-19 symptom burden: what is long-COVID and how should we manage it?
Lung.
(2021) 199:113–9. 10.1007/s00408-021-00423-z
Pubmed Abstract
CrossRef
Google Scholar
35.
Taquet
M
Dercon
Q
Luciano
S
Geddes
JR
Husain
M
Harrison
PJ
. Incidence, co-occurrence, and evolution of long-COVID features: A 6-month retrospective cohort study of 273,618 survivors of COVID-19.
PLoS Med.
(2021) 18:e1003773. 10.1371/journal.pmed.1003773
Pubmed Abstract
CrossRef
Google Scholar
36.
Tawfik
HM
Shaaban
HM
Tawfik
AM
. Post-COVID-19 syndrome in Egyptian healthcare staff: highlighting the carers sufferings.
Electron J Gen Med.
(2021) 18:em291. 10.29333/ejgm/10838
CrossRef
Google Scholar
37.
Morin
L
. The writing committee for the COMEBAC study group: four-month clinical status of a cohort of patients after hospitalization for COVID-19.
JAMA.
(2021) 325:1525–34. 10.1001/jama.2021.3331
Pubmed Abstract
CrossRef
Google Scholar
38.
van den Borst
B
Peters
JB
Brink
M
Schoon
Y
Bleeker-Rovers
CP
Schers
H
et al
. Comprehensive health assessment 3 months after recovery from acute coronavirus disease 2019 (COVID-19).
Clin Infect Dis Off Publ Infect Dis Soc Am.
(2021) 73:e1089–98. 10.1093/cid/ciaa1750
Pubmed Abstract
CrossRef
Google Scholar
39.
Vanichkachorn
G
Newcomb
R
Cowl
CT
Murad
MH
Breeher
L
Miller
S
et al
. Post–COVID-19 syndrome (Long Haul Syndrome): description of a multidisciplinary clinic at mayo clinic and characteristics of the initial patient cohort.
Mayo Clin Proc.
(2021) 96:1782–91. 10.1016/j.mayocp.2021.04.024
Pubmed Abstract
CrossRef
Google Scholar
40.
Vannorsdall
TD
Brigham
E
Fawzy
A
Raju
S
Gorgone
A
Pletnikova
A
et al
. Cognitive dysfunction, psychiatric distress, and functional decline after COVID-19.
J Acad Consult-Liaison Psychiatry.
(2022) 63:133–43. 10.1016/j.jaclp.2021.10.006
Pubmed Abstract
CrossRef
Google Scholar
41.
Vassalini
P
Serra
R
Tarsitani
L
Koukopoulos
AE
Borrazzo
C
Alessi
F
et al
. Depressive symptoms among individuals hospitalized with COVID-19: three-month follow-up.
Brain Sci.
(2021) 11:1175. 10.3390/brainsci11091175
Pubmed Abstract
CrossRef
Google Scholar
42.
CDC C for DC P
.
Subjective Cognitive Decline—A Public Health Issue.
(2019). Available online at:
https://www.cdc.gov/aging/data/subjective-cognitive-decline-brief.html
(accessed December 22, 2022).
Google Scholar
43.
Dattani
S
Ritchie
H
Roser
M
.
Mental Health. Our World Data.
(2021). Available online at:
https://ourworldindata.org/mental-health
(accessed December 22, 2022).
Google Scholar
44.
NIMH NI of MH
. Post-Traumatic Stress Disorder (PTSD).
Natl Inst Ment Health NIMH.
(2022) Available online at:
https://www.nimh.nih.gov/health/statistics/post-traumatic-stress-disorder-ptsd
(accessed December 22, 2022).
Google Scholar
45.
Stickley
A
Leinsalu
M
DeVylder
JE
Inoue
Y
Koyanagi
A
. Sleep problems and depression among 237 023 community-dwelling adults in 46 low- and middle-income countries.
Sci Rep.
(2019) 9:12011. 10.1038/s41598-019-48334-7
Pubmed Abstract
CrossRef
Google Scholar
46.
WHO WHO
.
Depression.
(2022). Available online at:
https://www.who.int/news-room/fact-sheets/detail/depression
(accessed December 22, 2022).
Google Scholar
47.
Hatch
R
Young
D
Barber
V
Griffiths
J
Harrison
DA
Watkinson
P
. Anxiety, depression and post traumatic stress disorder after critical illness: a UK-wide prospective cohort study.
Crit Care Lond Engl.
(2018) 22:310. 10.1186/s13054-018-2223-6
Pubmed Abstract
CrossRef
Google Scholar
48.
Benke
C
Autenrieth
LK
Asselmann
E
Pané-Farré
CA
. Lockdown, quarantine measures, and social distancing: Associations with depression, anxiety and distress at the beginning of the COVID-19 pandemic among adults from Germany.
Psychiatry Res.
(2020) 293:113462. 10.1016/j.psychres.2020.113462
Pubmed Abstract
CrossRef
Google Scholar
49.
Mastroberardino
M
Cuoghi Costantini
R
De Novellis
AMP
Ferrari
S
Filippini
C
Longo
F
et al
. “It's All COVID's Fault!”: symptoms of distress among workers in an italian general hospital during the pandemic.
Int J Environ Res Public Health.
(2022) 19:7313. 10.3390/ijerph19127313
Pubmed Abstract
CrossRef
Google Scholar
50.
Vindegaard
N
Benros
ME
. COVID-19 pandemic and mental health consequences: systematic review of the current evidence.
Brain Behav Immun.
(2020) 89:531–42. 10.1016/j.bbi.2020.05.048
Pubmed Abstract
CrossRef
Google Scholar
51.
Loades
ME
Chatburn
E
Higson-Sweeney
N
Reynolds
S
Shafran
R
Brigden
A
et al
. Rapid systematic review: the impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19.
J Am Acad Child Adolesc Psychiatr.
(2020) 59:1218-1239. 10.1016/j.jaac.2020.05.009
Pubmed Abstract
CrossRef
Google Scholar
52.
Marchi
M
Magarini
FM
Chiarenza
A
Galeazzi
GM
Paloma
V
Garrido
R
et al
. Experience of discrimination during COVID-19 pandemic: the impact of public health measures and psychological distress among refugees and other migrants in Europe.
BMC Public Health.
(2022) 22:942. 10.1186/s12889-022-13370-y
Pubmed Abstract
CrossRef
Google Scholar
53.
Choutka
J
Jansari
V
Hornig
M
Iwasaki
A
. Unexplained post-acute infection syndromes.
Nat Med.
(2022) 28:911–23. 10.1038/s41591-022-01810-6
Pubmed Abstract
CrossRef
Google Scholar
54.
Rogers
JP
Chesney
E
Oliver
D
Pollak
TA
McGuire
P
Fusar-Poli
P
et al
. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic.
Lancet Psychiatry.
(2020) 7:611–27. 10.1016/S2215-0366(20)30203-0
Pubmed Abstract
CrossRef
Google Scholar
55.
Balcom
EF
Nath
A
Power
C
. Acute and chronic neurological disorders in COVID-19: potential mechanisms of disease.
Brain J Neurol.
(2021) 144:3576–88. 10.1093/brain/awab302
Pubmed Abstract
CrossRef
Google Scholar
56.
Evans
RA
Leavy
OC
Richardson
M
Elneima
O
McAuley
HJC
Shikotra
A
et al
. Clinical characteristics with inflammation profiling of long COVID and association with 1-year recovery following hospitalisation in the UK: a prospective observational study.
Lancet Respir Med.
(2022) 10:761–75. 10.1016/S2213-2600(22)00127-8
Pubmed Abstract
CrossRef
Google Scholar
57.
Proal
AD
VanElzakker
MB
. Long COVID or Post-acute Sequelae of COVID-19 (PASC): An overview of biological factors that may contribute to persistent symptoms.
Front Microbiol.
(2021) 12:698169. 10.3389/fmicb.2021.698169
Pubmed Abstract
CrossRef
Google Scholar
58.
Theoharides
TC
Kempuraj
D
. Role of SARS-CoV-2 spike-protein-induced activation of microglia and mast cells in the pathogenesis of neuro-COVID.
Cells.
(2023) 12:688. 10.3390/cells12050688
Pubmed Abstract
CrossRef
Google Scholar
59.
Weinstock
LB
Brook
JB
Walters
AS
Goris
A
Afrin
LB
Molderings
GJ
. Mast cell activation symptoms are prevalent in Long-COVID.
Int J Infect Dis.
(2021) 112:217–26. 10.1016/j.ijid.2021.09.043
Pubmed Abstract
CrossRef
Google Scholar
60.
Song
S-T
Wu
M-L
Zhang
H-J
Su
X
Wang
J-H
. Mast cell activation triggered by retrovirus promotes acute viral infection.
Front Microbiol.
(2022) 13:8660. 10.3389/fmicb.2022.798660
Pubmed Abstract
CrossRef
Google Scholar
61.
Abraham
SN
John
AL
. Mast cell-orchestrated immunity to pathogens.
Nat Rev Immunol.
(2010) 10:440–52. 10.1038/nri2782
Pubmed Abstract
CrossRef
Google Scholar
62.
Hickie
I
Davenport
T
Wakefield
D
Vollmer-Conna
U
Cameron
B
Vernon
SD
et al
. Dubbo infection outcomes study group, Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study.
BMJ.
(2006) 333:575. 10.1136/bmj.38933.585764.AE
Pubmed Abstract
CrossRef
Google Scholar
63.
Hunskar
GS
Rortveit
G
Litleskare
S
Eide
GE
Hanevik
K
Langeland
N
et al
. Prevalence of fibromyalgia 10 years after infection with Giardia lamblia: a controlled prospective cohort study.
Scand J Pain.
(2022) 22:348–55. 10.1515/sjpain-2021-0122
Pubmed Abstract
CrossRef
Google Scholar
64.
Patel
H
Sander
B
Nelder
MP
. Long-term sequelae of West Nile virus-related illness: a systematic review.
Lancet Infect Dis.
(2015) 15:951–9. 10.1016/S1473-3099(15)00134-6
Pubmed Abstract
CrossRef
Google Scholar
65.
Rodríguez-Morales
AJ
Cardona-Ospina
JA
Fernanda Urbano-Garzón
S
Sebastian Hurtado-Zapata
J
. Prevalence of post-chikungunya infection chronic inflammatory arthritis: a systematic review and meta-analysis.
Arthritis Care Res.
(2016) 68:1849–58. 10.1002/acr.22900
Pubmed Abstract
CrossRef
Google Scholar
66.
The PREVAIL III
. Study group: a longitudinal study of Ebola Sequelae in Liberia.
N Engl J Med.
(2019) 380:924–34. 10.1056/NEJMoa1805435
Pubmed Abstract
CrossRef
Google Scholar
67.
White
PD
Thomas
JM
Amess
J
Crawford
DH
Grover
SA
Kangro
HO
et al
. Incidence, risk and prognosis of acute and chronic fatigue syndromes and psychiatric disorders after glandular fever.
Br J Psychiatry J Ment Sci.
(1998) 173:475–81. 10.1192/bjp.173.6.475
Pubmed Abstract
CrossRef
Google Scholar
68.
Macpherson
K
Cooper
K
Harbour
J
Mahal
D
Miller
C
Nairn
M
. Experiences of living with long COVID and of accessing healthcare services: a qualitative systematic review.
BMJ Open.
(2022) 12:e050979. 10.1136/bmjopen-2021-050979
Pubmed Abstract
CrossRef
Google Scholar
69.
Porter
N
Jason
LA
. Mindfulness meditation interventions for long COVID: biobehavioral gene expression and neuroimmune functioning.
Neuropsychiatr Dis Treat.
(2022) 18:2599–626. 10.2147/NDT.S379653
Pubmed Abstract
CrossRef
Google Scholar
Keywords
Long-COVID syndrome, COVID-19, mental health, depression, anxiety, posttraumatic stress
Citation
Marchi M, Grenzi P, Serafini V, Capoccia F, Rossi F, Marrino P, Pingani L, Galeazzi GM and Ferrari S (2023) Psychiatric symptoms in Long-COVID patients: a systematic review.
Front. Psychiatry
14:1138389. doi:
10.3389/fpsyt.2023.1138389
Received
05 January 2023
Accepted
29 May 2023
Published
21 June 2023
Volume
14 - 2023
Edited by
Zonglin He, Hong Kong University of Science and Technology, Hong Kong SAR, China
Reviewed by
Theoharis Constantin Theoharides, Nova Southeastern University, United States; Renata Kochhann, Hospital Moinhos de Vento, Brazil; Māris Taube, Riga Stradinš University, Latvia; Alberto Spalice, Sapienza University of Rome, Italy
Updates
Copyright
© 2023 Marchi, Grenzi, Serafini, Capoccia, Rossi, Marrino, Pingani, Galeazzi and Ferrari.
This is an open-access article distributed under the terms of the
Creative Commons Attribution License (CC BY)
. The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*
Correspondence:
Gian Maria Galeazzi
gianmaria.galeazzi@ausl.re.it
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher. | ||||||||||||||||||
| Markdown | [![]() Frontiers in Psychiatry](https://www.frontiersin.org/journals/psychiatry)
About us
About us
- Who we are
- [Mission and values](https://www.frontiersin.org/about/mission)
- [History](https://www.frontiersin.org/about/history)
- [Leadership](https://www.frontiersin.org/about/leadership)
- [Awards](https://www.frontiersin.org/about/awards)
- Impact and progress
- [Frontiers' impact](https://www.frontiersin.org/about/impact)
- [Our annual reports](https://www.frontiersin.org/about/annual-reports)
- [Thought leadership](https://www.frontiersin.org/about/thought-leadership)
- Publishing model
- [How we publish](https://www.frontiersin.org/about/how-we-publish)
- [Open access](https://www.frontiersin.org/about/open-access)
- [Quality and research integrity](https://www.frontiersin.org/about/quality)
- [Peer review](https://www.frontiersin.org/about/peer-review)
- [Research Topics](https://www.frontiersin.org/about/research-topics)
- [Publish your data](https://www.frontiersin.org/about/fair-data-management)
- [Fee policy](https://www.frontiersin.org/about/fee-policy)
- Services
- [Societies](https://publishingpartnerships.frontiersin.org/)
- [National consortia](https://www.frontiersin.org/open-access-agreements/consortia)
- [Institutional partnerships](https://www.frontiersin.org/about/open-access-agreements)
- [Collaborators](https://www.frontiersin.org/about/collaborators)
- More from Frontiers
- [Frontiers Forum](https://forum.frontiersin.org/)
- [Frontiers Planet Prize](https://www.frontiersin.org/about/frontiers-planet-prize)
- [Press office](https://pressoffice.frontiersin.org/)
- [Sustainability](https://www.frontiersin.org/about/sustainability)
- [Career opportunities](https://careers.frontiersin.org/)
- [Contact us](https://www.frontiersin.org/about/contact)
[All journals](https://www.frontiersin.org/journals)[All articles](https://www.frontiersin.org/articles)[Submit your research](https://www.frontiersin.org/submission/submit?domainid=2&fieldid=68&specialtyid=0&entitytype=2&entityid=71)
[Search](https://www.frontiersin.org/search?tab=top-results&origin=https%3A%2F%2Fwww.frontiersin.org%2Fjournals%2Fpsychiatry%2Farticles%2F10.3389%2Ffpsyt.2023.1138389%2Ffull)[Login](https://www.frontiersin.org/people/login?returnUrl=https%3A%2F%2Fwww.frontiersin.org%2Fjournals%2Fpsychiatry%2Farticles%2F10.3389%2Ffpsyt.2023.1138389%2Ffull)
[![]() Frontiers in Psychiatry](https://www.frontiersin.org/journals/psychiatry)
Sections
Sections
- [ADHD](https://www.frontiersin.org/journals/psychiatry/sections/adhd)
- [Addictive Disorders](https://www.frontiersin.org/journals/psychiatry/sections/addictive-disorders)
- [Adolescent and Young Adult Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/adolescent-and-young-adult-psychiatry)
- [Aging Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/aging-psychiatry)
- [Anxiety and Stress Disorders](https://www.frontiersin.org/journals/psychiatry/sections/anxiety-and-stress-disorders)
- [Autism](https://www.frontiersin.org/journals/psychiatry/sections/autism)
- [Behavioral and Psychiatric Genetics](https://www.frontiersin.org/journals/psychiatry/sections/behavioral-and-psychiatric-genetics)
- [Computational Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/computational-psychiatry)
- [Digital Mental Health](https://www.frontiersin.org/journals/psychiatry/sections/digital-mental-health)
- [Forensic Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/forensic-psychiatry)
- [Intellectual Disabilities](https://www.frontiersin.org/journals/psychiatry/sections/intellectual-disabilities)
- [Mental Health Occupational Therapy](https://www.frontiersin.org/journals/psychiatry/sections/mental-health-occupational-therapy)
- [Molecular Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/molecular-psychiatry)
- [Mood Disorders](https://www.frontiersin.org/journals/psychiatry/sections/mood-disorders)
- [Neuroimaging](https://www.frontiersin.org/journals/psychiatry/sections/neuroimaging)
- [Neurostimulation](https://www.frontiersin.org/journals/psychiatry/sections/neurostimulation)
- [Perinatal Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/perinatal-psychiatry)
- [Personality Disorders](https://www.frontiersin.org/journals/psychiatry/sections/personality-disorders)
- [Psychological Therapy and Psychosomatics](https://www.frontiersin.org/journals/psychiatry/sections/psychological-therapy-and-psychosomatics)
- [Psychopathology](https://www.frontiersin.org/journals/psychiatry/sections/psychopathology)
- [Psychopharmacology](https://www.frontiersin.org/journals/psychiatry/sections/psychopharmacology)
- [Public Mental Health](https://www.frontiersin.org/journals/psychiatry/sections/public-mental-health)
- [Schizophrenia](https://www.frontiersin.org/journals/psychiatry/sections/schizophrenia)
- [Sleep Disorders](https://www.frontiersin.org/journals/psychiatry/sections/sleep-disorders)
- [Social Neuroscience](https://www.frontiersin.org/journals/psychiatry/sections/social-neuroscience)
- [Social Psychiatry and Psychiatric Rehabilitation](https://www.frontiersin.org/journals/psychiatry/sections/social-psychiatry-and-psychiatric-rehabilitation)
[Articles](https://www.frontiersin.org/journals/psychiatry/articles)[Research Topics](https://www.frontiersin.org/journals/psychiatry/research-topics)[Editorial board](https://www.frontiersin.org/journals/psychiatry/editors)
About journal
About journal
- Scope
- [Field chief editors](https://www.frontiersin.org/journals/psychiatry/about#about-editors)
- [Mission & scope](https://www.frontiersin.org/journals/psychiatry/about#about-scope)
- [Facts](https://www.frontiersin.org/journals/psychiatry/about#about-facts)
- [Journal sections](https://www.frontiersin.org/journals/psychiatry/about#about-submission)
- [Open access statement](https://www.frontiersin.org/journals/psychiatry/about#about-open)
- [Copyright statement](https://www.frontiersin.org/journals/psychiatry/about#copyright-statement)
- [Quality](https://www.frontiersin.org/journals/psychiatry/about#about-quality)
- For authors
- [Why submit?](https://www.frontiersin.org/journals/psychiatry/for-authors/why-submit)
- [Article types](https://www.frontiersin.org/journals/psychiatry/for-authors/article-types)
- [Author guidelines](https://www.frontiersin.org/journals/psychiatry/for-authors/author-guidelines)
- [Editor guidelines](https://www.frontiersin.org/journals/psychiatry/for-authors/editor-guidelines)
- [Publishing fees](https://www.frontiersin.org/journals/psychiatry/for-authors/publishing-fees)
- [Submission checklist](https://www.frontiersin.org/journals/psychiatry/for-authors/submission-checklist)
- [Contact editorial office](https://www.frontiersin.org/journals/psychiatry/for-authors/contact-editorial-office)
About us
About us
- Who we are
- [Mission and values](https://www.frontiersin.org/about/mission)
- [History](https://www.frontiersin.org/about/history)
- [Leadership](https://www.frontiersin.org/about/leadership)
- [Awards](https://www.frontiersin.org/about/awards)
- Impact and progress
- [Frontiers' impact](https://www.frontiersin.org/about/impact)
- [Our annual reports](https://www.frontiersin.org/about/annual-reports)
- [Thought leadership](https://www.frontiersin.org/about/thought-leadership)
- Publishing model
- [How we publish](https://www.frontiersin.org/about/how-we-publish)
- [Open access](https://www.frontiersin.org/about/open-access)
- [Quality and research integrity](https://www.frontiersin.org/about/quality)
- [Peer review](https://www.frontiersin.org/about/peer-review)
- [Research Topics](https://www.frontiersin.org/about/research-topics)
- [Publish your data](https://www.frontiersin.org/about/fair-data-management)
- [Fee policy](https://www.frontiersin.org/about/fee-policy)
- Services
- [Societies](https://publishingpartnerships.frontiersin.org/)
- [National consortia](https://www.frontiersin.org/open-access-agreements/consortia)
- [Institutional partnerships](https://www.frontiersin.org/about/open-access-agreements)
- [Collaborators](https://www.frontiersin.org/about/collaborators)
- More from Frontiers
- [Frontiers Forum](https://forum.frontiersin.org/)
- [Frontiers Planet Prize](https://www.frontiersin.org/about/frontiers-planet-prize)
- [Press office](https://pressoffice.frontiersin.org/)
- [Sustainability](https://www.frontiersin.org/about/sustainability)
- [Career opportunities](https://careers.frontiersin.org/)
- [Contact us](https://www.frontiersin.org/about/contact)
[All journals](https://www.frontiersin.org/journals)[All articles](https://www.frontiersin.org/articles)[Submit your research](https://www.frontiersin.org/submission/submit?domainid=2&fieldid=68&specialtyid=0&entitytype=2&entityid=71)
[![]() Frontiers in Psychiatry](https://www.frontiersin.org/journals/psychiatry)
Sections
Sections
- [ADHD](https://www.frontiersin.org/journals/psychiatry/sections/adhd)
- [Addictive Disorders](https://www.frontiersin.org/journals/psychiatry/sections/addictive-disorders)
- [Adolescent and Young Adult Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/adolescent-and-young-adult-psychiatry)
- [Aging Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/aging-psychiatry)
- [Anxiety and Stress Disorders](https://www.frontiersin.org/journals/psychiatry/sections/anxiety-and-stress-disorders)
- [Autism](https://www.frontiersin.org/journals/psychiatry/sections/autism)
- [Behavioral and Psychiatric Genetics](https://www.frontiersin.org/journals/psychiatry/sections/behavioral-and-psychiatric-genetics)
- [Computational Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/computational-psychiatry)
- [Digital Mental Health](https://www.frontiersin.org/journals/psychiatry/sections/digital-mental-health)
- [Forensic Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/forensic-psychiatry)
- [Intellectual Disabilities](https://www.frontiersin.org/journals/psychiatry/sections/intellectual-disabilities)
- [Mental Health Occupational Therapy](https://www.frontiersin.org/journals/psychiatry/sections/mental-health-occupational-therapy)
- [Molecular Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/molecular-psychiatry)
- [Mood Disorders](https://www.frontiersin.org/journals/psychiatry/sections/mood-disorders)
- [Neuroimaging](https://www.frontiersin.org/journals/psychiatry/sections/neuroimaging)
- [Neurostimulation](https://www.frontiersin.org/journals/psychiatry/sections/neurostimulation)
- [Perinatal Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/perinatal-psychiatry)
- [Personality Disorders](https://www.frontiersin.org/journals/psychiatry/sections/personality-disorders)
- [Psychological Therapy and Psychosomatics](https://www.frontiersin.org/journals/psychiatry/sections/psychological-therapy-and-psychosomatics)
- [Psychopathology](https://www.frontiersin.org/journals/psychiatry/sections/psychopathology)
- [Psychopharmacology](https://www.frontiersin.org/journals/psychiatry/sections/psychopharmacology)
- [Public Mental Health](https://www.frontiersin.org/journals/psychiatry/sections/public-mental-health)
- [Schizophrenia](https://www.frontiersin.org/journals/psychiatry/sections/schizophrenia)
- [Sleep Disorders](https://www.frontiersin.org/journals/psychiatry/sections/sleep-disorders)
- [Social Neuroscience](https://www.frontiersin.org/journals/psychiatry/sections/social-neuroscience)
- [Social Psychiatry and Psychiatric Rehabilitation](https://www.frontiersin.org/journals/psychiatry/sections/social-psychiatry-and-psychiatric-rehabilitation)
[Articles](https://www.frontiersin.org/journals/psychiatry/articles)[Research Topics](https://www.frontiersin.org/journals/psychiatry/research-topics)[Editorial board](https://www.frontiersin.org/journals/psychiatry/editors)
About journal
About journal
- Scope
- [Field chief editors](https://www.frontiersin.org/journals/psychiatry/about#about-editors)
- [Mission & scope](https://www.frontiersin.org/journals/psychiatry/about#about-scope)
- [Facts](https://www.frontiersin.org/journals/psychiatry/about#about-facts)
- [Journal sections](https://www.frontiersin.org/journals/psychiatry/about#about-submission)
- [Open access statement](https://www.frontiersin.org/journals/psychiatry/about#about-open)
- [Copyright statement](https://www.frontiersin.org/journals/psychiatry/about#copyright-statement)
- [Quality](https://www.frontiersin.org/journals/psychiatry/about#about-quality)
- For authors
- [Why submit?](https://www.frontiersin.org/journals/psychiatry/for-authors/why-submit)
- [Article types](https://www.frontiersin.org/journals/psychiatry/for-authors/article-types)
- [Author guidelines](https://www.frontiersin.org/journals/psychiatry/for-authors/author-guidelines)
- [Editor guidelines](https://www.frontiersin.org/journals/psychiatry/for-authors/editor-guidelines)
- [Publishing fees](https://www.frontiersin.org/journals/psychiatry/for-authors/publishing-fees)
- [Submission checklist](https://www.frontiersin.org/journals/psychiatry/for-authors/submission-checklist)
- [Contact editorial office](https://www.frontiersin.org/journals/psychiatry/for-authors/contact-editorial-office)
[![]() Frontiers in Psychiatry](https://www.frontiersin.org/journals/psychiatry)
Sections
Sections
- [ADHD](https://www.frontiersin.org/journals/psychiatry/sections/adhd)
- [Addictive Disorders](https://www.frontiersin.org/journals/psychiatry/sections/addictive-disorders)
- [Adolescent and Young Adult Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/adolescent-and-young-adult-psychiatry)
- [Aging Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/aging-psychiatry)
- [Anxiety and Stress Disorders](https://www.frontiersin.org/journals/psychiatry/sections/anxiety-and-stress-disorders)
- [Autism](https://www.frontiersin.org/journals/psychiatry/sections/autism)
- [Behavioral and Psychiatric Genetics](https://www.frontiersin.org/journals/psychiatry/sections/behavioral-and-psychiatric-genetics)
- [Computational Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/computational-psychiatry)
- [Digital Mental Health](https://www.frontiersin.org/journals/psychiatry/sections/digital-mental-health)
- [Forensic Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/forensic-psychiatry)
- [Intellectual Disabilities](https://www.frontiersin.org/journals/psychiatry/sections/intellectual-disabilities)
- [Mental Health Occupational Therapy](https://www.frontiersin.org/journals/psychiatry/sections/mental-health-occupational-therapy)
- [Molecular Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/molecular-psychiatry)
- [Mood Disorders](https://www.frontiersin.org/journals/psychiatry/sections/mood-disorders)
- [Neuroimaging](https://www.frontiersin.org/journals/psychiatry/sections/neuroimaging)
- [Neurostimulation](https://www.frontiersin.org/journals/psychiatry/sections/neurostimulation)
- [Perinatal Psychiatry](https://www.frontiersin.org/journals/psychiatry/sections/perinatal-psychiatry)
- [Personality Disorders](https://www.frontiersin.org/journals/psychiatry/sections/personality-disorders)
- [Psychological Therapy and Psychosomatics](https://www.frontiersin.org/journals/psychiatry/sections/psychological-therapy-and-psychosomatics)
- [Psychopathology](https://www.frontiersin.org/journals/psychiatry/sections/psychopathology)
- [Psychopharmacology](https://www.frontiersin.org/journals/psychiatry/sections/psychopharmacology)
- [Public Mental Health](https://www.frontiersin.org/journals/psychiatry/sections/public-mental-health)
- [Schizophrenia](https://www.frontiersin.org/journals/psychiatry/sections/schizophrenia)
- [Sleep Disorders](https://www.frontiersin.org/journals/psychiatry/sections/sleep-disorders)
- [Social Neuroscience](https://www.frontiersin.org/journals/psychiatry/sections/social-neuroscience)
- [Social Psychiatry and Psychiatric Rehabilitation](https://www.frontiersin.org/journals/psychiatry/sections/social-psychiatry-and-psychiatric-rehabilitation)
[Articles](https://www.frontiersin.org/journals/psychiatry/articles)[Research Topics](https://www.frontiersin.org/journals/psychiatry/research-topics)[Editorial board](https://www.frontiersin.org/journals/psychiatry/editors)
About journal
About journal
- Scope
- [Field chief editors](https://www.frontiersin.org/journals/psychiatry/about#about-editors)
- [Mission & scope](https://www.frontiersin.org/journals/psychiatry/about#about-scope)
- [Facts](https://www.frontiersin.org/journals/psychiatry/about#about-facts)
- [Journal sections](https://www.frontiersin.org/journals/psychiatry/about#about-submission)
- [Open access statement](https://www.frontiersin.org/journals/psychiatry/about#about-open)
- [Copyright statement](https://www.frontiersin.org/journals/psychiatry/about#copyright-statement)
- [Quality](https://www.frontiersin.org/journals/psychiatry/about#about-quality)
- For authors
- [Why submit?](https://www.frontiersin.org/journals/psychiatry/for-authors/why-submit)
- [Article types](https://www.frontiersin.org/journals/psychiatry/for-authors/article-types)
- [Author guidelines](https://www.frontiersin.org/journals/psychiatry/for-authors/author-guidelines)
- [Editor guidelines](https://www.frontiersin.org/journals/psychiatry/for-authors/editor-guidelines)
- [Publishing fees](https://www.frontiersin.org/journals/psychiatry/for-authors/publishing-fees)
- [Submission checklist](https://www.frontiersin.org/journals/psychiatry/for-authors/submission-checklist)
- [Contact editorial office](https://www.frontiersin.org/journals/psychiatry/for-authors/contact-editorial-office)
[Submit your research](https://www.frontiersin.org/submission/submit?domainid=2&fieldid=68&specialtyid=0&entitytype=2&entityid=71)[Search](https://www.frontiersin.org/search?tab=top-results&origin=https%3A%2F%2Fwww.frontiersin.org%2Fjournals%2Fpsychiatry%2Farticles%2F10.3389%2Ffpsyt.2023.1138389%2Ffull)[Login](https://www.frontiersin.org/people/login?returnUrl=https%3A%2F%2Fwww.frontiersin.org%2Fjournals%2Fpsychiatry%2Farticles%2F10.3389%2Ffpsyt.2023.1138389%2Ffull)
## SYSTEMATIC REVIEW article
Front. Psychiatry, 21 June 2023
Sec. Public Mental Health
Volume 14 - 2023 \| <https://doi.org/10.3389/fpsyt.2023.1138389>
## SYSTEMATIC REVIEW article
Front. Psychiatry, 21 June 2023
Sec. Public Mental Health
Volume 14 - 2023 \| <https://doi.org/10.3389/fpsyt.2023.1138389>
# Psychiatric symptoms in Long-COVID patients: a systematic review
- [MMMattia Marchi 1,2](https://loop.frontiersin.org/people/1782154)
- PG
Pietro Grenzi 1
- VS
Valentina Serafini 1
- FC
Francesco Capoccia 1
- FR
Federico Rossi 1
- PM
Patrizia Marrino 1
- [LPLuca Pingani 1,2](https://loop.frontiersin.org/people/494437)
- [GMGian Maria Galeazzi 1,2\*](https://loop.frontiersin.org/people/598018)
- [SFSilvia Ferrari 1,2](https://loop.frontiersin.org/people/615345)
- 1\. Department of Biomedical, Metabolic and Neural Sciences, University of Modena and Reggio Emilia, Modena, Italy
- 2\. Department of Mental Health and Drug Abuse, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy
Article metrics
[View details](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#metrics)
62
Citations
8,3k
Views
2,1k
Downloads
## Abstract
**Objective:**
People who have been infected by COVID-19 showing persistent symptoms after 4 weeks from recovery are thought to suffer from Long-COVID syndrome (LC). There is uncertainty on the clinical manifestations of LC. We undertook a systematic review to summarize the available evidence about the main psychiatric manifestations of LC.
**Method:**
PubMed (Medline), Scopus, CINHAL, PsycINFO, and EMBASE were searched until May 2022. Studies reporting estimation of emerging psychiatric symptoms and/or psychiatric diagnoses among adult people with LC were included. Pooled prevalence for each psychiatric condition was calculated in absence of control groups to compare with.
**Results:**
Thirty-three reports were included in the final selection, corresponding to 282,711 participants with LC. After 4 weeks from COVID-19 infection recovery, participants reported the following psychiatric symptoms: depression, anxiety, post-traumatic symptoms (PTS), cognitive and sleeping disturbances (i.e., insomnia or hypersomnia). The most common psychiatric manifestation resulted to be sleep disturbances, followed by depression, PTS, anxiety, and cognitive impairment (i.e., attention and memory deficits). However, some estimates were affected by important outlier effect played by one study. If study weight was not considered, the most reported condition was anxiety.
**Conclusions:**
LC may have non-specific psychiatric manifestations. More research is needed to better define LC and to differentiate it from other post-infectious or post-hospitalization syndromes.
**Systematic review registration:**
PROSPERO (CRD42022299408).
## Introduction
Long-COVID syndrome (LC) is a condition that can affect people who have recovered from Coronavirus Disease 2019 (COVID-19). This term was introduced to indicate a set of disorders that persist or occur at from 4 weeks after the elimination of the SARS-CoV-2 virus from the body ([1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B1)). The clinical features of LC are multifaceted; it has been posited that it can affect different organs and systems, causing somatic but also psychological manifestations that impact on quality of life ([2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B2)).
For most people, mild or moderate COVID-19 lasts for about 2 weeks; in some cases, though, symptoms can persist or develop after healing. Furthermore, also in people with asymptomatic infections later health problems may develop ([3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B3)–[5](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B5)).
Although progress has been made in the understanding of the clinical and epidemiological features, including the pathogenesis and complications of the acute phase of COVID-19, long-term consequences of the disease remain largely unclear ([6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6)).
Additionally, while neuropsychiatric symptoms that manifest acutely during infection, such as depression, post-traumatic symptoms \[PTS\], sleep and cognitive disturbances or anxiety, have received more attention, the medium- and long-term psychiatric outcomes in COVID-19 patients are still little known and understudied ([7](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B7), [8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8)).
In the available literature, there are highly heterogeneous research works on this topic, applying widely different sample sizes, inclusion and exclusion criteria, and duration of follow-up. In addition, patient assessment is mainly based on various assessment tools and questionnaires, self-administered in most cases, that do not provide a diagnosis of a condition with definite clinical significance.
Therefore, understanding the medium and long-term impact of COVID-19 is still far from being complete, not only in the context of a multidisciplinary approach, but even more so when focusing on specific areas such as mental health ([1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B1)).
We undertook this systematic review to summarize the available evidence about the main psychiatric manifestations of LC. A better understanding of the epidemiology of psychopathological manifestations among LC patients is crucial to develop prevention and early interventions.
## Methods
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol of this systematic review was registered with PROSPERO (CRD42022299408).
### Data sources and search strategy
We searched the PubMed (Medline), Scopus, CINHAL, PsycINFO, and EMBASE databases until May 2022, using the strategy outlined in the [Supplementary Table 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1) of the [Appendix](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1). In addition, the list of references of the included studies and of other reviews on related topics was screened to identify any other possible study deserving inclusion, and inadvertently missed during the initial literature search. No restrictions regarding language of publication or publication date were set.
### Eligibility criteria
We included experimental and observational studies reporting estimation of rates of emerging psychiatric symptoms and/or psychiatric diagnosis among adult people (i.e., ≥18 years old) with LC, without any restriction on other medical comorbidities or setting of enrolment. We excluded studies on participants already suffering from any psychiatric condition, studies assessing the presence of psychiatric symptoms before 4 weeks from COVID-19 recovery, and previous reviews, case-reports, case-series, editorial, and letters to the editor. We only included studies published in peer-reviewed journals, excluding conference abstracts and dissertations. If data from the same sample were published in multiple works, we considered only that study reporting more exhaustive information. Sample overlap was ruled out through a careful check of the registration codes as well as the place and year(s) of sampling.
Where available, outcome data from participants with other inflammatory or infectious diseases, including COVID-19 but without LC, were used as control group.
### Terms and definitions
LC was defined as either the presence or the persistence of any symptom that was not present before the infection after 4 weeks from the COVID-19 recovery. Infection from SARS-CoV-2 and recovery from the infection were defined according to the result of the real-time PCR on nasopharyngeal swab sample, or of broncho-alveolar lavage.
Psychiatric symptoms were collected from self-reporting or from validated psychometric tools. Where a psychiatric diagnosis was reported, it had to be defined according to standard operational diagnostic criteria (the Diagnostic and Statistical Manual of Mental Disorders \[DSM\] or the International Classification of Diseases \[ICD\]).
### Data collection and extraction
Four Reviewers (P.G., V.S., F.R., and F.C.) working independently preliminarily reviewed titles and abstracts of retrieved articles. The initial screening was followed by the analysis of full texts to check compliance with inclusion/exclusion criteria. All disagreements were discussed until consensus, and if consensus was not possible, another member of the team was consulted (M.M.). A standardized form was used for data extraction. Information concerning the year of publication, country, setting, characteristics of study participants (sample size, age, percentages of men and women), LC status, and the presence of psychiatric conditions in the LC groups (and, where available, in the control group) were collected by two authors (P.G. and V.S.) independently. Extraction sheets for each study were cross-checked for consistency and any disagreement was resolved by discussion within the research group.
### Statistical analyses
Where possible (i.e., there were at least two studies providing outcome data for LC and controls), quantitative data among studies were summarized using random effects meta-analysis ([9](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B9)). To summarize continuous outcome data (i.e., the scores on a psychometric tool), the pooled Hedges' g standardized mean differences (SMDs) and the corresponding 95% confidence intervals (CIs) were applied, while pooled odds ratios (ORs) and the corresponding 95% CIs ([10](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B10)) were used to report on dichotomous outcome data (i.e., presence/absence of psychiatric diagnosis or psychiatric symptoms).
If meta-analysis was not possible, we calculated the pooled prevalence of psychiatric symptoms and/or psychiatric diagnosis among LC patients. These estimates consisted in weighted-mean prevalence, raw mean prevalence, and median prevalence, with the relative lower and upper ranges across the studies included in the final selection.
The analyses were performed in R ([11](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B11)). Statistical tests were 2-sided and used a significance threshold of *p* \< 0.05.
### Risk of bias assessment and the GRADE
Bias risk in the included studies was independently assessed by three reviewers (P.G., V.S., and F.R.), using the Cochrane risk of bias tool ([12](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B12)). All disagreements were discussed until consensus, and if necessary, another member of the team was consulted (M.M.). Each item on the risk of bias assessment was scored as high, low, or unclear, and the GRADE tool was used to assess the overall certainty of evidence ([13](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B13)). Further information is available in the [Supplementary material](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1).
## Results
### Study characteristics
As shown in [Figure 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F1), from 2078 records screened on title and abstract, 114 full texts were analyzed. The review process led to the selection of 33 studies ([3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B3), [4](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B4), [6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6)–[8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8), [14](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B14)–[41](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B41)). These studies, referring to 33 different samples and involving a total of 282,711 LC participants, were included in the final selection and quantitative synthesis.
Figure 1

Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram.
On an average, across the studies, 48% of participants were females (range 23.1–100%). The mean age of participants was 53.2 years (range 33.7–73.2). The selected studies were conducted in 13 countries: US (*n* = 6; 18.2%); Italy (*n* = 5; 15.2%); Egypt, France, Netherlands, Spain, UK (each *n* = 3; 9.1%); India (*n* = 2; 6.1%); Austria, China, Germany, Iran, Mexico (each *n* = 1; 3.0%).
All the studies were published in the last 2 years: 2021 (*n* = 31; 93.9%); 2022 (*n* = 2; 6.1%).
With respect to the outcomes reported, only 2 studies (6.1%) provided data about psychiatric diagnosis: one study assessed depressive and anxiety disorder (GAD) through a clinical interview, the other study used retrospective screening of the electronic clinical records to investigate prevalence of anxiety and depression, and cognitive impairment, according to the ICD-10 system. The remaining studies (*n* = 31, 93.9%) used self-reporting or other psychometric tools to measure the level of: depression or anxiety (*n* = 26, 78.8%), cognitive impairment (*n* = 16, 48.5%), PTS (*n* = 13, 39.4%), and sleep disturbances (*n* = 18, 54.5%). These studies applied dichotomization into positive/negative at the psychometric assessment based on the scales' cut-off for clinical significance, and the estimated prevalence for each study was calculated as the number of participants with score above the cut-off divided to the total number of participants assessed.
Notably, none of the studies included in the final selection applied a control group without LC. Concerning the severity of COVID-19 infection, 15 studies (45.5%) included patients hospitalized due to COVID-19 infection, 5 studies (15.2%) were performed on patients who had mild infection not requiring hospitalization, and 11 studies (33.3%) included both hospitalized and other managed outpatients. Information about infection severity was missing in 2 studies (6.1%).
All studies characteristics are summarized in [Table 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T1).
Table 1
| **Author, year** | **Country** | **Study design** | **Females %** | **Mean age** | **N** | **Severity of COVID-19 infection** | **P depression (measure)** | **P Anxiety (measure)** | **P Cognitive impairment (measure)** | **P PTS (measure)** | **P Sleep disturbances (measure)** |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ahmed et al. ([14](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B14)) | Egypt | Cohort | 54% | 46\.5 | 182 | Both hospitalized and outpatients | 0\.374 (SCL-90) | 0\.619 (SCL-90) | NR | 0\.286 (PCL-5) | 0\.648 (PSQI) |
| Aly and Saber ([15](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B15)) | Egypt | Cross-sectional | 100% | 73\.2 | 115 | NR | NR | NR | 0\.252 (self-reported) | NR | 0\.243 (self-reported) |
| Aranda et al. ([16](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B16)) | Spain | Cohort | 30% | 64 | 113 | Hospitalized | 0\.301 (BDI) | 0\.487 (STAI) | NR | 0\.788 (IES-R) | 0\.292 (NR) |
| Bai et al. ([17](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B17)) | Italy | Cohort | 36% | 57 | 377 | Both hospitalized and outpatients | 0\.106 (HADS) | 0\.188 (HADS) | 0\.202 (NR) | 0\.225 (IES-R) | NR |
| Boesl et al. ([18](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B18)) | Italy | Cohort | 67% | 45\.8 | 100 | Mild | 0\.615 (BDI) | NR | 0\.306 (MoCA) | NR | 0\.337 (ESS) |
| De Graaf et al. ([3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B3)) | Netherlands | Cohort | 37% | 60\.8 | 81 | Both hospitalized and outpatients | 0\.123 (PHQ-9) | 0\.037 (GAD-7) | 0\.160 (CFQ-25) | 0\.062 (PCL-5) | NR |
| Evans et al. ([4](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B4)) | UK | Cohort | 36% | 57\.9 | 1077 | Hospitalized | 0\.262 (PHQ-9) | 0\.235 (GAD-7) | 0\.139 (NR) | 0\.117 (PCL-5) | NR |
| Frontera et al. ([19](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B19)) | US | Case-control | 35% | 68\.5 | 280 | Hospitalized | 0\.254 (NeuroQoL) | 0\.511 (NeuroQoL) | 0\.473 (MoCA) | NR | 0\.375 (NeuroQoL) |
| Ganesh et al. ([20](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B20)) | US | Cohort | 61% | 44 | 817 | Both hospitalized and outpatients | NR | NR | NR | NR | 0\.200 (PROMIS) |
| Garjani et al. ([21](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B21)) | UK | Cohort | 82% | 50 | 165 | Mild | 0\.504 (NR)† | NR | NR | NR | NR |
| Gonzàlez-Hermosillo et al. ([22](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B22)) | Mexico | Cohort | 35% | 51 | 130 | Hospitalized | 0\.354 (self-reported) | 0\.392 (self-reported) | 0\.454 (self-reported) | NR | 0\.454 (self-reported) |
| Gouraud et al. ([23](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B23)) | France | Cohort | 29% | 60 | 100 | Hospitalized | 0\.220 (HADS) | 0\.310 (HADS) | NR | NR | NR |
| Graham et al. ([24](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B24)) | US | Case-control | 66% | 43\.7 | 50 | Mild | 0\.400 (NR)† | NR | 0\.820 (PROMIS) | NR | 0\.360 (PROMIS) |
| Gramaglia et al. ([6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6)) | Italy | Cohort | 40% | 61 | 238 | Both hospitalized and outpatients | 0\.294 (MINI) | 0\.328 (MINI) | NR | 0\.429 (NR) | NR |
| Horwitz et al. ([7](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B7)) | US | Cohort | 40% | 62 | 126 | Hospitalized | NR | NR | 0\.413 (PROMIS) | NR | 0\.349 (PROMIS) |
| Huang et al. ([8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8)) | China | Cohort | 48% | 57 | 1733 | Hospitalized | 0\.227 (interview)† | NR | NR | NR | 0\.264 (interview) |
| Imran et al. ([25](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B25)) | India | Cross-sectional | 33% | 44\.5 | 103 | Hospitalized | 0\.126 (PHQ-9) | 0\.214 (GAD-7) | NR | 0\.087 (PCL-5) | NR |
| Lemhofer et al. ([26](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B26)) | Germany | Cross-sectional | 59% | 49\.8 | 365 | Mild | NR | 0\.249 (NR) | NR | NR | 0\.301 (NR) |
| Lombardo et al. ([27](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B27)) | Italy | Cohort | 54% | 53 | 303 | Both hospitalized and outpatients | NR | NR | 0\.363 (semi-structured interview) | NR | 0\.465 (semi-structured interview) |
| Mendez et al. ([28](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B28)) | Spain | Cohort | 41% | 57 | 179 | Hospitalized | 0\.268 (PHQ-9) | 0\.296 (GAD-7) | 0\.184 (NR) | 0\.251 (DTS) | NR |
| Morin et al. ([37](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B37)) | France | Cohort | 42% | 60\.9 | 478 | Hospitalized | 0\.206 (BDI) | 0\.314 (HADS) | 0\.384 (MoCA, D2-R, Q3PC) | 0\.142 (PCL-5) | 0\.536 (ISI) |
| Naik et al. ([29](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B29)) | India | Cohort | 31% | 41\.6 | 272 | Both hospitalized and outpatients | 0\.022 (interview) | 0\.029 (interview) | NR | NR | 0\.063 (interview) |
| Rass et al. ([30](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B30)) | Austria | Cohort | 39% | 55 | 90 | Both hospitalized and outpatients | 0\.121 (HADS) | 0\.222 (HADS) | NR | 0\.100 (PCL-5) | NR |
| Romero-Duarte et al. ([31](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B31)) | Spain | Cohort | 46% | 63 | 794 | Hospitalized | 0\.044 (NR) | NR | NR | NR | 0\.049 (NR) |
| Scherlinger et al. ([32](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B32)) | France | Case-control | 66% | 40 | 30 | Mild | 0\.100 (Psychological interview) | 0\.267 (Psychological interview) | NR | 0\.300 (PCL-5) | NR |
| Simani et al. ([33](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B33)) | Iran | Cohort | 33% | 54\.62 | 120 | Hospitalized | NR | NR | NR | 0\.058 (PCL-5) | NR |
| Sykes et al., ([34](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B34)) | UK | Cohort | 34% | | 134 | Hospitalized | 0\.396 (self-reported) | 0\.478 (self-reported) | 0\.097 (self-reported) | NR | 0\.351 (self-reported) |
| Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) | Netherlands | Cohort | 58% | 39\.4 | 273618 | Both hospitalized and outpatients | 0\.155 (ICD-10)† | NR | 0\.040 (ICD-10) | NR | NR |
| Tawfik ([36](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B36)) | Egypt | Cohort | 58% | 33\.7 | 120 | Both hospitalized and outpatients | NR | NR | 0\.008 (NR) | NR | 0\.042 (NR) |
| Van den Borst et al. ([38](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B38)) | Netherlands | Cohort | 40% | 59 | 124 | Both hospitalized and outpatients | 0\.117 (HADS) | 0\.100 (HADS) | NR | 0\.073 (PCL-5) | NR |
| Vanichkachorn et al. ([39](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B39)) | US | Cohort | 68% | 45\.7 | 100 | NR | NR | NR | 0\.005 (NR) | NR | 0\.003 (NR) |
| Vannorsdall et al. ([40](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B40)) | US | Cohort | 59% | 54\.5 | 82 | Hospitalized | 0\.902 (PHQ-9) | 0\.646 (GAD-7) | 0\.805 (QDRS) | 0\.25 (IES-R) | NR |
| Vassalini et al. ([41](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B41)) | Italy | Cohort | 46% | 57 | 115 | Hospitalized | 0\.148 (PHQ-9) | NR | NR | NR | NR |
Characteristics of the included studies.
P, prevalence; PTS, post-traumatic symptoms; NR, not-reported; SCL-90, symptom checklist 90; PCL-5, PTSD checklist for DSM-5; PSQI, Pittsburgh sleep quality index; BDI, Beck depression inventory; STAI, state-trait anxiety inventory; IES-R, impact of event scale—revised; HADS, hospital anxiety and depression scale; MoCA, Montreal cognitive assessment; ESS, Epworth sleepiness scale; PHQ-9, patient health questionnaire-9; GAD-7, general anxiety disorder-7; CFQ-25, cognitive failure questionnaire-25; UK, United Kingdom; US, United States of America; NeuroQoL, health-related quality of life for clinical research in neurology; PROMIS, patient-reported outcomes measurement information system; MINI, mini-international neuropsychiatric interview; DTS, Davidson trauma scale; D2-R, D2 test of attention—revised; Q3PC, Q3PC cognitive screening questionnaire; ISI, insomnia severity index; ICD-10, international classification of diseases 10th revision; QDRS, quick dementia rating scale.
†depression and anxiety aggregated prevalence.
### Prevalence of psychiatric symptoms across the studies
[Table 2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T2) summarize the pooled prevalence estimates for each psychiatric symptom across the studies included in this review, and the population prevalence worldwide. Notably, prevalence of depression, anxiety, cognitive impairment, PTS, and sleep disturbances resulted much higher among LC patients than in the general population ([42](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B42)–[46](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B46)).
Table 2
| **Outcome** | **Weighted mean P** | **Mean P (min; max)** | **Median P** | **N studies (LC participants)** | **World P** |
|---|---|---|---|---|---|
| Depression | 0\.212 | 0\.254 (0.022; 0.902) | 0\.220 | 21 (5,079) | 0\.038 |
| Anxiety | 0\.158 | 0\.313 (0.029; 0.646) | 0\.296 | 23 (28,001) | 0\.040 |
| Cognitive impairment | 0\.042 | 0\.269 (0.005; 0.820) | 0\.227 | 16 (277,268) | 0\.011 |
| PTS | 0\.192 | 0\.218 (0.058; 0.788) | 0\.130 | 13 (3,162) | 0\.036 |
| Sleeping disturbances | 0\.270 | 0\.296 (0.003; 0.648) | 0\.319 | 18 (6,212) | 0\.038 |
Pooled prevalence of psychiatric symptoms across the included studies and worldwide prevalence.
P, prevalence; min, minimum; max, maximum; LC, Long-COVID syndrome; NA, not available; PTS, post-traumatic symptoms.
### Prevalence of depression
Twenty-one studies (63.3%) provided outcome data for depression among LC patients. The weighted mean prevalence across the studies was 0.212, that is quite similar to the unweighted mean and median prevalence (0.254 \[range: 0.022–0.902\] and 0.220, respectively), consistent with not significant outlier effect played by any of the study in the pooled estimate. [Figure 2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F2) shows comparison of the depression prevalence estimates across the studies, and the weighted mean prevalence.
Figure 2
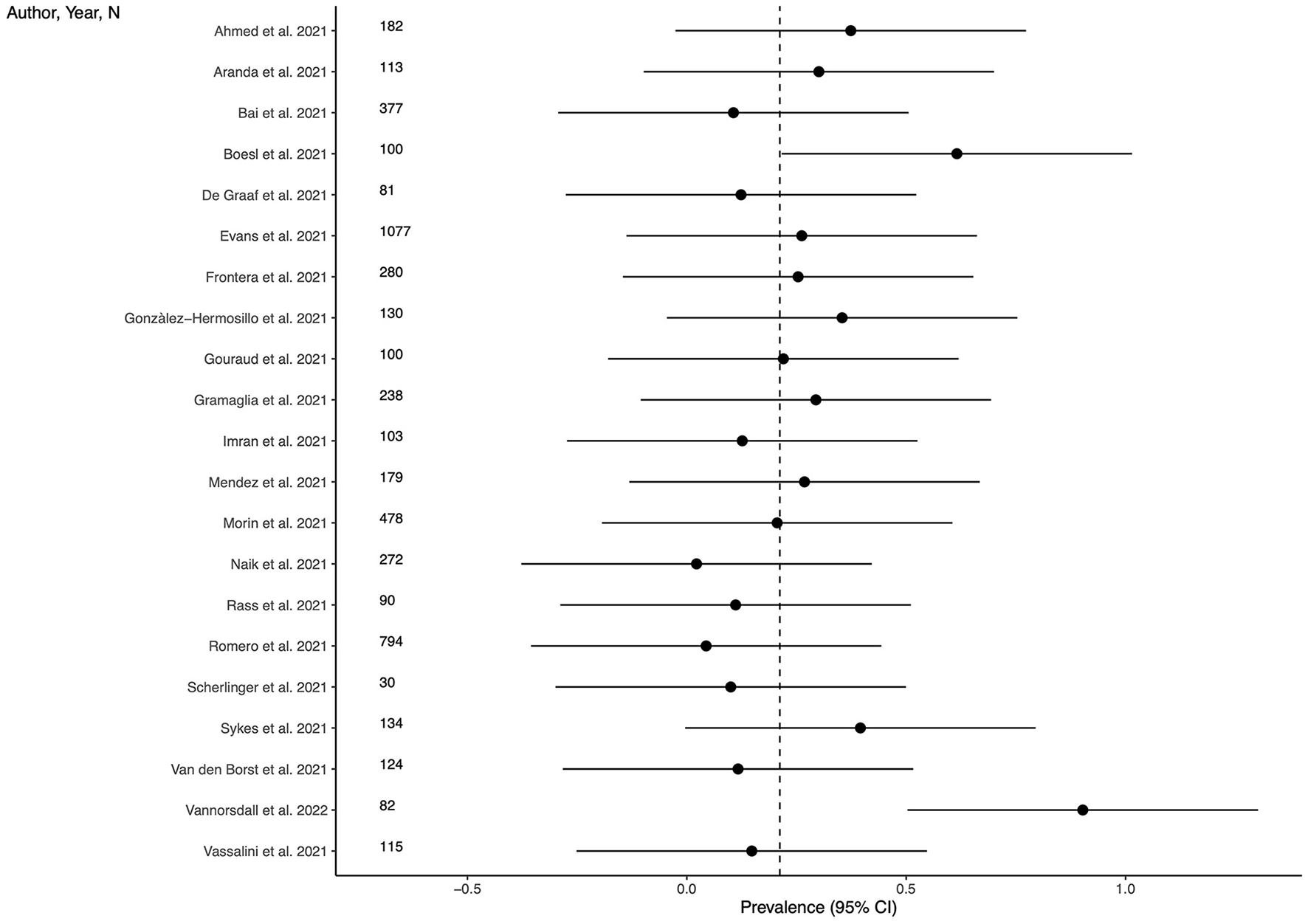
Prevalence of depression across the studies and weighted mean prevalence. The vertical dotted line represents the weighted mean prevalence.
### Prevalence of anxiety
Twenty-three studies (69.7%) provided outcome data for anxiety among LC patients. The weighted mean prevalence across the studies was 0.158 and was markedly influenced by the study from Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) with a far larger sample size. Unweighted mean and median prevalence were 0.313 (range: 0.029–0.646) and 0.296, respectively. [Figure 3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F3) shows comparison of the anxiety prevalence estimates across the studies, and the weighted mean prevalence.
Figure 3

Prevalence of anxiety across the studies and weighted mean prevalence. The vertical dotted line represents the weighted mean prevalence.
### Prevalence of cognitive impairment
Sixteen studies (48.5%) provided outcome data for cognitive impairment among LC patients. The weighted mean prevalence across the studies was 0.042 and, again, was markedly influenced by the study from Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) with the largest sample size and providing among the three lowest estimates of anxiety prevalence. Unweighted mean and median prevalence were 0.269 (range: 0.005–0.820) and 0.227, respectively. [Figure 4](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F4) shows comparison of the cognitive impairment prevalence estimates across the studies, and the weighted mean prevalence.
Figure 4
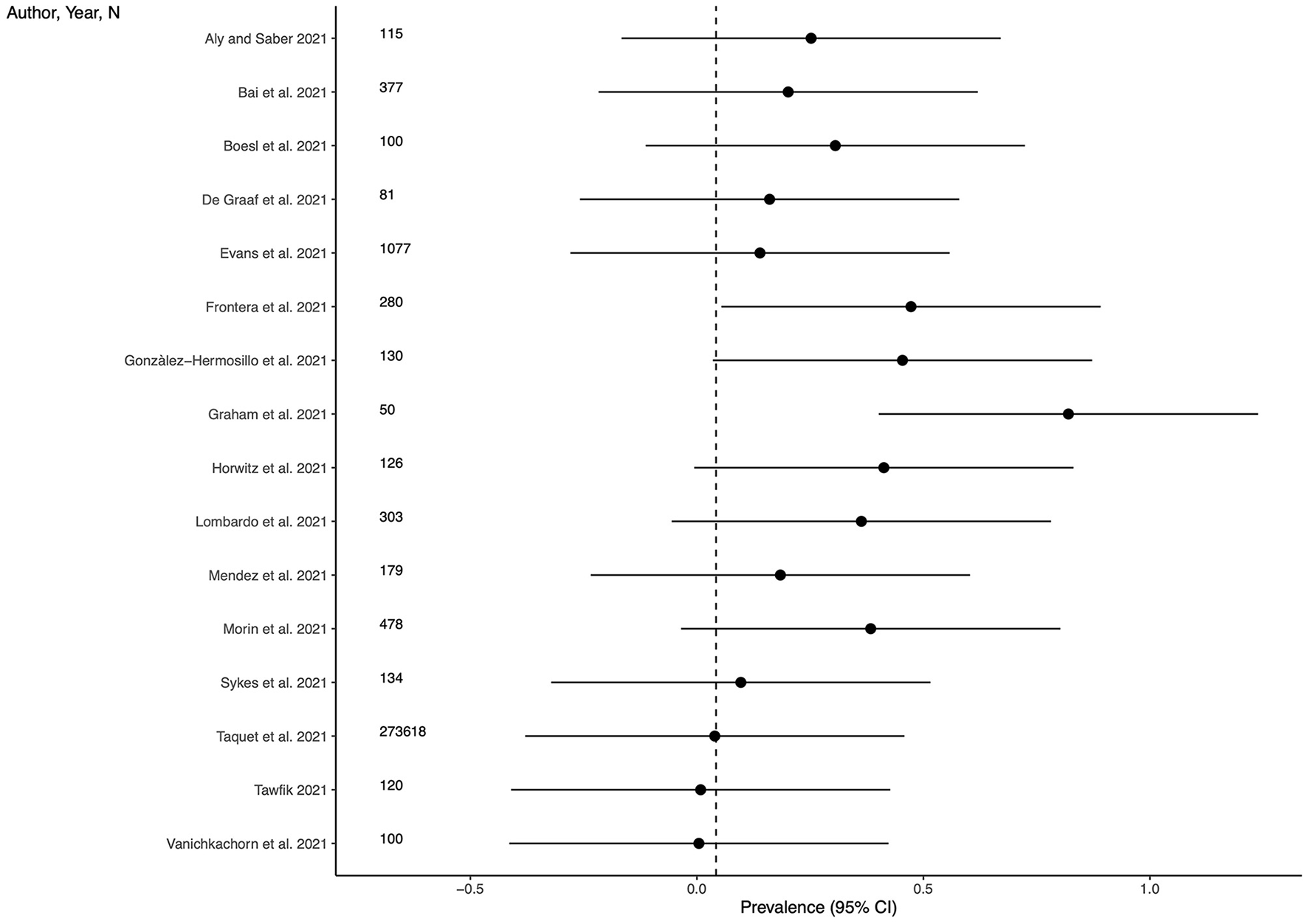
Prevalence of cognitive impairment across the studies and weighted mean prevalence. The vertical dotted line represents the weighted mean prevalence.
### Prevalence of PTS
Thirteen studies (39.4%) provided outcome data for PTS among LC patients. The weighted mean prevalence across the studies was 0.192. Unweighted mean and median prevalence were 0.218 (range: 0.058–0.788) and 0.130, respectively. [Figure 5](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F5) shows comparison of the PTS prevalence estimates across the studies, and the weighted mean prevalence.
Figure 5

Prevalence of post-traumatic symptoms across the studies and weighted mean prevalence. The vertical dotted line represents the weighted mean prevalence.
### Prevalence of sleep disturbances
Eighteen studies (54.5%) provided outcome data for sleep disturbances among LC patients. The weighted mean prevalence across the studies was 0.270. Unweighted mean and median prevalence were 0.296 (range: 0.003–0.648) and 0.319, respectively. [Figure 6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F6) shows comparison of the sleep disturbances prevalence estimates across the studies, and the weighted mean prevalence.
Figure 6

Prevalence of sleep disturbances across the studies and weighted mean prevalence. The vertical dotted line represents the weighted mean prevalence.
### Psychiatric symptoms by COVID-19 infection severity
We examined the potential association between the severity of COVID-19 infection and the occurrence of psychiatric symptoms. For this purpose, we considered hospitalization as a proxy for severe infection, and outpatient management as indicator of mild infection. As shown in [Table 3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T3), the comparison of the severe, mild, and both mild and severe groups in terms of average prevalence of psychiatric symptoms did not find any statistically significant difference, suggesting that the severity of the infection is not related to the development of later psychiatric symptoms. Further analysis was conducted to compare only mild and severe patients, with the severe group consisting of studies that included at least one hospitalized patient. This analysis confirmed that there were no statistically significant differences in depression, anxiety, PTS, and sleep disturbances, but found inverse association between the severity of the infection and cognitive complaints (*p* = 0.048).
Table 3
| | **Mean P (SD) mild** | **Mean P (SD) severe** | **Mean P (SD) both** | ***p*\-value** |
|---|---|---|---|---|
| N | 5 | 15 | 11 | |
| Depression | 0\.36 (0.36) | 0\.29 (0.22) | 0\.16 (0.12) | 0\.339 |
| Anxiety | 0\.36 (0.12) | 0\.37 (0.14) | 0\.21 (0.19) | 0\.089 |
| Cognitive impairment | 0\.56 (0.36) | 0\.31 (0.16) | 0\.15 (0.14) | 0\.061 |
| PTS | NA | 0\.24 (0.28) | 0\.20 (0.15) | 0\.731 |
| Sleeping disturbances | 0\.33 (0.03) | 0\.33 (0.14) | 0\.28 (0.26) | 0\.879 |
Prevalence of psychiatric symptoms by severity of the COVID-19 infection.
P, prevalence; SD, standard deviation; N, number; PTS, post-traumatic symptoms; NA, not available.
p-value are based on multiple groups analysis of variance (ANOVA).
### Risk of bias and GRADE
A detailed summary on the risk of bias in all 33 trials has been reported in the [Appendix](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1) (see [Supplementary Figures 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1), [2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1)), along with an assessment of the quality of the evidence (see [Supplementary Table 2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1)). In the GRADE system, the evidence from observational studies is initially set to low, there are then criteria that can be used either to downgrade or upgrade (see further information in the [Appendix](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1)). The quality of the evidence is rated very low with serious threats related to the risk of bias and inconsistency.
## Discussion
This study set out to investigate the prevalence of psychiatric symptoms among LC patients. We found that the symptoms mostly associated with LC were depression, anxiety, cognitive and sleep disturbances, and PTS. The prevalence of these symptoms among LC patients is remarkably higher than that in the general population.
However, it is necessary to underline that, among the studies included in the final selection, there was one study ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) that had a sample size accounting for around 96% of the total number of participants included in this review. That study provided estimates for anxiety and cognitive impairments and the weighted average for these outcomes falls exactly on the value of the prevalence estimated by Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) by looking at the forest plots of anxiety and cognitive impairment, it can be easily observed that only three studies for anxiety and two studies for cognitive impairment provided estimates smaller than Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) supporting its influence on the pooled prevalence. Accordingly, for these two outcomes, the raw mean resulted higher than the weighted mean, because the former is not affected by the differences in the sample size across the studies.
Nevertheless, the relatively high pooled prevalence of psychiatric symptoms among LC patients requires a better understanding. Our analyses did not find a significant association between the severity of COVID-19 infection and psychiatric symptoms, except for a potential inverse association with symptoms of cognitive impairment. These findings align with the conclusions drawn from most of the reports included in our systematic review, which examined the relationship between infection severity and psychiatric symptoms ([6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6), [17](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B17), [20](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B20), [23](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B23), [27](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B27), [34](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B34), [40](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B40)). Notably, only one study reported an increased risk of depression and anxiety among individuals with the most severe form of infection ([8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8)). However, it is important to acknowledge that the confidence in the results is limited by the comparatively small representation of patients with mild infection, leading to low statistical power.
It should be noted that research in the pre-COVID-19 era observed that survivors after intensive care (IC) are at greater risk of developing long-term mental disorders ([47](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B47)). Particularly, anxiety, depression, and PTSD would have occurred in half of this sample of UK patients discharged from IC. Psychiatric symptoms of the post-IC syndrome fall into three broad categories: physical, cognitive, and psychological deterioration. Symptoms of physical deterioration include fatigue and insomnia, while cognitive and psychiatric symptoms include anxiety, depression, memory impairment, and PTSD. Therefore, there seems to be a significant overlap in the experience of some of the LC patients analyzed in this review with post-IC syndrome.
A considerable amount of COVID-related research also focused on the effect on mental health of public health measures (such as quarantine, lock-down, social isolation and other limitations to personal freedom), finding an association with symptoms of depression, anxiety, loneliness, psychosocial distress, and persisting post-traumatic arousal ([48](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B48), [49](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B49)). Therefore, another possible explanation for the higher prevalence of psychiatric symptoms among LC patients may be more a consequence of the imposed quarantine and other restrictions in terms of anxiety, fear, anger, and other negative emotions, regardless of specific aspects of the COVID-19 infection such as neuro- or systemic inflammation. Even if the quarantine imposed by a local health authority has not been directly associated with any psychological outcomes ([50](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B50)), it was suggested that belonging to a publicly recognized COVID-19 risk group/community would be associated with increased anxiety, depressive symptoms, self-concern, fear, increased psychosocial distress, and decreased life satisfaction. In addition, loneliness and isolation have been associated with an increased risk for various mental disorders (as well as for various somatic diseases). In the context of the COVID-19 pandemic, loneliness has been found to be predictive of depressive and anxious symptoms during the lockdown measures ([51](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B51), [52](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B52)).
COVID-19 is in fact only the most recent of many other infectious diseases that have been associated with chronic sequelae after recovering from the acute phase of infection ([53](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B53)): similarly as with LC, the underlying pathophysiological and etiological mechanisms are far from being clearly understood. The review by Choutka et al. ([53](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B53)) investigated the common characteristics between LC and other chronic infectious syndromes, finding higher prevalence of the following symptoms: intolerance to physical effort, neurocognitive and sensory impairment, persistent flu-like symptoms, disturbed sleep, myalgias, and arthralgias. The greatest analogies are with the post-acute effects described in the SARS epidemic in 2002–2004 ([54](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B54)).
All these elements are probably interrelated with each other and influential in the experience of LC patients.
### Limitations
The results of this review should be interpreted considering its limitations. First, the lack of a control group made difficult to draw considerations on the risk of psychiatric symptoms among LC patients, reducing considerably generalizability and reliability of our findings. This translated also in the impossibility to meta-analyze the results of the selected studies to detail the risk of psychiatric symptoms in patients who have had COVID-19. Second, most of the included studies reported measures of psychiatric symptoms instead of assessing psychiatric diagnoses, with a risk for diagnostic overestimation: this was partially attenuated by including only studies applying validated psychometric tools or clinical interviews. Third, there were a marked outlier effect played by one study ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)), which implemented a sample size accounting for more than 90% of the total sample size. Even though that study resulted at low risk of bias in the assessment, it may have impacted on the pooled prevalence estimate of the outcomes reported in that study. Fourth, the condition of LC has been assessed only through a temporal criterion, that was 4 weeks after recovery from the infection. The lack of a more comprehensive definition of LC may have increase the heterogeneity in the estimates. Finally, the risk of bias was rated high or unclear in many studies, with serious threats related to the inconsistency in the estimates and to the assessment of confounders.
### Implications for research and clinical practice
The overlapping of some clinical features of LC in terms of signs and symptoms with other post-infectious syndromes and with the post-IC syndrome would suggest the involvement of shared pathophysiological pathways. The perspective of identifying a unified etiological model would lead the way toward the implementation of diagnostic markers and tailored treatments ([53](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B53)). At present, however, our understanding of the underlying pathophysiological mechanisms and etiological factors is poor, though promising studies are being conducted ([55](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B55)–[57](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B57)). For example, a recent review advanced the hypothesis that perivascular inflammation serves as the critical pathogenetic factor for LC neuropsychiatric manifestations ([58](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B58)). Indeed, SARS-CoV-2 and other viruses (such as retrovirus) showed the ability to activate brain mast cells and microglia resulting in the release of inflammatory, neurotoxic, and vasoactive mediators impacting neuronal connectivity and signal transmission ([59](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B59)–[61](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B61)). Hopefully, that may also converge to a better definition of functional and psycho-somatic syndromes, such as fibromyalgia and chronic fatigue syndrome, for which the association with viral infections has been previously proposed ([62](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B62)–[67](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B67)).
More research is therefore needed, more clearly comparing different patient groups (e.g., LC patients that were admitted to ICU vs. other ICU patients with or without other infectious diseases; LC patients vs. patients remitting from other infectious diseases) and applying prospective designs, allowing causal considerations, and providing more epidemiological details. Also, qualitative studies investigating the subjective experience of people recovered from COVID-19 are being conducted ([68](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B68)): this approach may also contribute to the understanding of the psychological mechanism contributing to the onset of psychological symptoms. Such different research methods could converge on a better conceptualization and analysis of the symptoms associated with the LC syndrome, as well as supporting the construction of a better defined and unified nomenclature.
A better understanding of the LC psycho-pathophysiology is essential to provide and improve treatment. From a therapeutic point of view, in close relation both to the traumatic component of a part of the symptoms found in LC, and to the inflammatory component (initially exerted by the infection and then self-sustained), interventions aimed at reducing the inflammatory process and reducing the excessive activation of the sympathetic nervous system through a relaxation response may be useful. For example, models of intervention involving reconditioning and mindfulness may help patients suffering from LC ([69](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B69)). Future clinical trials on LC patients may be therefore warranted.
## Conclusions
People who have recovered from COVID-19 may experience more and persistent psychiatric symptoms. These include depression, anxiety, post-traumatic distress, cognitive and sleeping disturbances. However, there is marked heterogeneity in the literature about how these symptoms are investigated and differentiated from other post-infectious or post-hospitalization conditions. More research, particularly implementing control groups and prospective follow-up, are needed to better define psychopathology related or included into the LC syndrome.
## Statements
### Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
### Author contributions
MM, PG, and VS: conceptualization and planning and interpretation of the results. PG, VS, FC, FR, and PM: acquisition. MM: analysis of the data. MM, PG, VS, FC, FR, and PM: drafting. SF, LP, and GG: critical revision of the manuscript. All authors approved the final submitted version of the manuscript.
### Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
### Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
### Supplementary material
The Supplementary Material for this article can be found online at: [https://www.frontiersin.org/articles/10.3389/fpsyt.2023.1138389/full\#supplementary-material](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#supplementary-material)
## References
- 1\.
AkbarialiabadHTaghrirMHAbdollahiAGhahramaniNKumarMPaydarSet al. Long COVID, a comprehensive systematic scoping review. *Infection.* (2021) 49:1163–86. 10.1007/s15010-021-01666-x
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34319569)
- [CrossRef](https://doi.org/10.1007/s15010-021-01666-x)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=H.%2BAkbarialiabad&author=MH.%2BTaghrir&author=A.%2BAbdollahi&author=N.%2BGhahramani&author=M.%2BKumar&author=S.%2BPaydar&publication_year=2021&title=Long%2BCOVID%2C%2Ba%2Bcomprehensive%2Bsystematic%2Bscoping%2Breview&journal=Infection.&volume=49&pages=1163-86)
- 2\.
MichelenMManoharanLElkheirNChengVDagensAHastieCet al. Characterising long COVID: a living systematic review. *BMJ Glob Health.* (2021) 6:e005427. 10.1136/bmjgh-2021-005427
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34580069)
- [CrossRef](https://doi.org/10.1136/bmjgh-2021-005427)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BMichelen&author=L.%2BManoharan&author=N.%2BElkheir&author=V.%2BCheng&author=A.%2BDagens&author=C.%2BHastie&publication_year=2021&title=Characterising%2Blong%2BCOVID%3A%2Ba%2Bliving%2Bsystematic%2Breview&journal=BMJ+Glob+Health.&volume=6)
- 3\.
De GraafMAAntoniMLKuileMMArbousMSDuinisveldAJFFeltkampMCWet al. Short-term outpatient follow-up of COVID-19 patients: a multidisciplinary approach. *eClinicalMedicine.* (2021) 32:25. 10.1016/j.eclinm.2021.100731
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33532720)
- [CrossRef](https://doi.org/10.1016/j.eclinm.2021.100731)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=MA.%2BDe%2BGraaf&author=ML.%2BAntoni&author=MM.%2BKuile&author=MS.%2BArbous&author=AJF.%2BDuinisveld&author=MCW.%2BFeltkamp&publication_year=2021&title=Short-term%2Boutpatient%2Bfollow-up%2Bof%2BCOVID-19%2Bpatients%3A%2Ba%2Bmultidisciplinary%2Bapproach&journal=eClinicalMedicine.&volume=32)
- 4\.
EvansRAMcAuleyHHarrisonEMShikotraASingapuriASerenoMet al. Physical, cognitive, and mental health impacts of COVID-19 after hospitalisation (PHOSP-COVID): a UK multicentre, prospective cohort study. *Lancet Respir Med.* (2021) 9:1275–87. 10.1016/S2213-2600(21)00383-0
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34627560)
- [CrossRef](https://doi.org/10.1016/S2213-2600\(21\)00383-0)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=RA.%2BEvans&author=H.%2BMcAuley&author=EM.%2BHarrison&author=A.%2BShikotra&author=A.%2BSingapuri&author=M.%2BSereno&publication_year=2021&title=Physical%2C%2Bcognitive%2C%2Band%2Bmental%2Bhealth%2Bimpacts%2Bof%2BCOVID-19%2Bafter%2Bhospitalisation%2B%28PHOSP-COVID%29%3A%2Ba%2BUK%2Bmulticentre%2C%2Bprospective%2Bcohort%2Bstudy&journal=Lancet+Respir+Med.&volume=9&pages=1275-87)
- 5\.
FioreGFerrariSCutinoAGiorginoCValeoLGaleazziGMet al. Delirium in COVID-19 and post-liver transplant patients: an observational study. *Int J Psychiatry Clin Pract.* (2022) 26:343–51. 10.1080/13651501.2022.2026403
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35061952)
- [CrossRef](https://doi.org/10.1080/13651501.2022.2026403)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=G.%2BFiore&author=S.%2BFerrari&author=A.%2BCutino&author=C.%2BGiorgino&author=L.%2BValeo&author=GM.%2BGaleazzi&publication_year=2022&title=Delirium%2Bin%2BCOVID-19%2Band%2Bpost-liver%2Btransplant%2Bpatients%3A%2Ban%2Bobservational%2Bstudy&journal=Int+J+Psychiatry+Clin+Pract.&volume=26&pages=343-51)
- 6\.
GramagliaCGambaroEBellanMBalboPEBaricichASainaghiPPet al. Mid-term psychiatric outcomes of patients recovered from COVID-19 from an italian cohort of hospitalized patients. *Front Psychiatry.* (2021) 12:667385. 10.3389/fpsyt.2021.667385
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34177656)
- [CrossRef](https://doi.org/10.3389/fpsyt.2021.667385)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BGramaglia&author=E.%2BGambaro&author=M.%2BBellan&author=PE.%2BBalbo&author=A.%2BBaricich&author=PP.%2BSainaghi&publication_year=2021&title=Mid-term%2Bpsychiatric%2Boutcomes%2Bof%2Bpatients%2Brecovered%2Bfrom%2BCOVID-19%2Bfrom%2Ban%2Bitalian%2Bcohort%2Bof%2Bhospitalized%2Bpatients&journal=Front+Psychiatry.&volume=12)
- 7\.
HorwitzLIGarryKPreteAMSharmaSMendozaFKahanTet al. Six-month outcomes in patients hospitalized with severe COVID-19. *J Gen Intern Med.* (2021) 36:3772–7. 10.1007/s11606-021-07032-9
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34355349)
- [CrossRef](https://doi.org/10.1007/s11606-021-07032-9)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=LI.%2BHorwitz&author=K.%2BGarry&author=AM.%2BPrete&author=S.%2BSharma&author=F.%2BMendoza&author=T.%2BKahan&publication_year=2021&title=Six-month%2Boutcomes%2Bin%2Bpatients%2Bhospitalized%2Bwith%2Bsevere%2BCOVID-19&journal=J+Gen+Intern+Med.&volume=36&pages=3772-7)
- 8\.
HuangCHuangLWangYLiXRenLGuXet al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. *Lancet.* (2021) 397:220–32. 10.1016/S0140-6736(20)32656-8
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/37307841)
- [CrossRef](https://doi.org/10.1016/S0140-6736\(20\)32656-8)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BHuang&author=L.%2BHuang&author=Y.%2BWang&author=X.%2BLi&author=L.%2BRen&author=X.%2BGu&publication_year=2021&title=6-month%2Bconsequences%2Bof%2BCOVID-19%2Bin%2Bpatients%2Bdischarged%2Bfrom%2Bhospital%3A%2Ba%2Bcohort%2Bstudy&journal=Lancet.&volume=397&pages=220-32)
- 9\.
DerSimonianRLairdN. Meta-analysis in clinical trials. *Control Clin Trials.* (1986) 7:177–88. 10.1016/0197-2456(86)90046-2
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/3802833)
- [CrossRef](https://doi.org/10.1016/0197-2456\(86\)90046-2)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BDerSimonian&author=N.%2BLaird&publication_year=1986&title=Meta-analysis%2Bin%2Bclinical%2Btrials&journal=Control+Clin+Trials.&volume=7&pages=177-88)
- 10\.
HigginsJThomasJChandlerJCumpstonMLiTPageM. *Cochrane Handbook for Systematic Reviews of Interventions. version 6.2. Cochrane* (2021). Available online at: [www.training.cochrane.org/handbook](http://www.training.cochrane.org/handbook). (accessed January 28, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BHiggins&author=J.%2BThomas&author=J.%2BChandler&author=M.%2BCumpston&author=T.%2BLi&author=M.%2BPage&publication_year=2021&journal=Cochrane+Handbook+for+Systematic+Reviews+of+Interventions.+version+6.2.+Cochrane)
- 11\.
RStudio Team. *RStudio: Integrated Development Environment for R.* (2021) Available online at: <http://www.rstudio.com/>
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2021&journal=RStudio%3A+Integrated+Development+Environment+for+R.)
- 12\.
HigginsJPTAltmanDGGøtzschePCJüniPMoherDOxmanADet al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. *BMJ.* (2011) 343:d5928. 10.1136/bmj.d5928
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/22008217)
- [CrossRef](https://doi.org/10.1136/bmj.d5928)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JPT.%2BHiggins&author=DG.%2BAltman&author=PC.%2BG%C3%B8tzsche&author=P.%2BJ%C3%BCni&author=D.%2BMoher&author=AD.%2BOxman&publication_year=2011&title=The%2BCochrane%2BCollaboration%27s%2Btool%2Bfor%2Bassessing%2Brisk%2Bof%2Bbias%2Bin%2Brandomised%2Btrials&journal=BMJ.&volume=343)
- 13\.
SchünemannHBrozekJGuyattGOxmanA. *GRADE Handbook for Grading Quality of Evidence Strength of Recommendations.* (2013). Available online at: <https://gdt.gradepro.org/app/handbook/handbook.html> (accessed July 6, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=H.%2BSch%C3%BCnemann&author=J.%2BBrozek&author=G.%2BGuyatt&author=A.%2BOxman&publication_year=2013&journal=GRADE+Handbook+for+Grading+Quality+of+Evidence+Strength+of+Recommendations.)
- 14\.
AhmedGKKhedrEMHamadDAMeshrefTSHashemMMAlyMM. Long term impact of Covid-19 infection on sleep and mental health: a cross-sectional study. *Psychiatry Res.* (2021) 305:114243. 10.1016/j.psychres.2021.114243
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34673325)
- [CrossRef](https://doi.org/10.1016/j.psychres.2021.114243)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=GK.%2BAhmed&author=EM.%2BKhedr&author=DA.%2BHamad&author=TS.%2BMeshref&author=MM.%2BHashem&author=MM.%2BAly&publication_year=2021&title=Long%2Bterm%2Bimpact%2Bof%2BCovid-19%2Binfection%2Bon%2Bsleep%2Band%2Bmental%2Bhealth%3A%2Ba%2Bcross-sectional%2Bstudy&journal=Psychiatry+Res.&volume=305)
- 15\.
AlyMAEGSaberHG. Long COVID and chronic fatigue syndrome: a survey of elderly female survivors in Egypt. *Int J Clin Pract.* (2021) 75:e14886. 10.1111/ijcp.14886
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34537995)
- [CrossRef](https://doi.org/10.1111/ijcp.14886)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=MAEG.%2BAly&author=HG.%2BSaber&publication_year=2021&title=Long%2BCOVID%2Band%2Bchronic%2Bfatigue%2Bsyndrome%3A%2Ba%2Bsurvey%2Bof%2Belderly%2Bfemale%2Bsurvivors%2Bin%2BEgypt&journal=Int+J+Clin+Pract.&volume=75)
- 16\.
ArandaJOriolIMartínMFeriaLVázquezNRhymanNet al. Long-term impact of COVID-19 associated acute respiratory distress syndrome. *J Infect.* (2021) 83:581–8. 10.1016/j.jinf.2021.08.018
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34400219)
- [CrossRef](https://doi.org/10.1016/j.jinf.2021.08.018)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BAranda&author=I.%2BOriol&author=M.%2BMart%C3%ADn&author=L.%2BFeria&author=N.%2BV%C3%A1zquez&author=N.%2BRhyman&publication_year=2021&title=Long-term%2Bimpact%2Bof%2BCOVID-19%2Bassociated%2Bacute%2Brespiratory%2Bdistress%2Bsyndrome&journal=J+Infect.&volume=83&pages=581-8)
- 17\.
BaiFTomasoniDFalcinellaCBarbanottiDCastoldiRMulèGet al. Female gender is associated with long COVID syndrome: a prospective cohort study. *Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis.* (2022) 28:611. 10.1016/j.cmi.2021.11.002
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34763058)
- [CrossRef](https://doi.org/10.1016/j.cmi.2021.11.002)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=F.%2BBai&author=D.%2BTomasoni&author=C.%2BFalcinella&author=D.%2BBarbanotti&author=R.%2BCastoldi&author=G.%2BMul%C3%A8&publication_year=2022&title=Female%2Bgender%2Bis%2Bassociated%2Bwith%2Blong%2BCOVID%2Bsyndrome%3A%2Ba%2Bprospective%2Bcohort%2Bstudy&journal=Clin+Microbiol+Infect+Off+Publ+Eur+Soc+Clin+Microbiol+Infect+Dis.&volume=28)
- 18\.
BoeslFAudebertHEndresMPrüssHFrankeC. A neurological outpatient clinic for patients with post-COVID-19 syndrome—a report on the clinical presentations of the first 100 patients. *Front Neurol.* (2021) 12:405. 10.3389/fneur.2021.738405
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34603189)
- [CrossRef](https://doi.org/10.3389/fneur.2021.738405)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=F.%2BBoesl&author=H.%2BAudebert&author=M.%2BEndres&author=H.%2BPr%C3%BCss&author=C.%2BFranke&publication_year=2021&title=A%2Bneurological%2Boutpatient%2Bclinic%2Bfor%2Bpatients%2Bwith%2Bpost-COVID-19%2Bsyndrome%E2%80%94a%2Breport%2Bon%2Bthe%2Bclinical%2Bpresentations%2Bof%2Bthe%2Bfirst%2B100%2Bpatients&journal=Front+Neurol.&volume=12)
- 19\.
FronteraJAYangDLewisAPatelPMedicherlaCArenaVet al. A prospective study of long-term outcomes among hospitalized COVID-19 patients with and without neurological complications. *J Neurol Sci.* (2021) 426:7486. 10.1016/j.jns.2021.117486
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34000678)
- [CrossRef](https://doi.org/10.1016/j.jns.2021.117486)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JA.%2BFrontera&author=D.%2BYang&author=A.%2BLewis&author=P.%2BPatel&author=C.%2BMedicherla&author=V.%2BArena&publication_year=2021&title=A%2Bprospective%2Bstudy%2Bof%2Blong-term%2Boutcomes%2Bamong%2Bhospitalized%2BCOVID-19%2Bpatients%2Bwith%2Band%2Bwithout%2Bneurological%2Bcomplications&journal=J+Neurol+Sci.&volume=426)
- 20\.
GaneshRGhoshAKNymanMACroghanITGrachSLAnstineCVet al. Scales for assessment of persistent post-COVID symptoms: a cross sectional study. *J Prim Care Community Health.* (2021) 12:21501327211030412. 10.1177/21501327211030413
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34231395)
- [CrossRef](https://doi.org/10.1177/21501327211030413)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BGanesh&author=AK.%2BGhosh&author=MA.%2BNyman&author=IT.%2BCroghan&author=SL.%2BGrach&author=CV.%2BAnstine&publication_year=2021&title=Scales%2Bfor%2Bassessment%2Bof%2Bpersistent%2Bpost-COVID%2Bsymptoms%3A%2Ba%2Bcross%2Bsectional%2Bstudy&journal=J+Prim+Care+Community+Health.&volume=12)
- 21\.
GarjaniAMiddletonRMNicholasREvangelouN. Recovery from COVID-19 in multiple sclerosis: a prospective and longitudinal cohort study of the united kingdom multiple sclerosis register. *Neurol—Neuroimmunol Neuroinflamm.* (2022) 9:1118. 10.1212/NXI.0000000000001118
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34848503)
- [CrossRef](https://doi.org/10.1212/NXI.0000000000001118)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=A.%2BGarjani&author=RM.%2BMiddleton&author=R.%2BNicholas&author=N.%2BEvangelou&publication_year=2022&title=Recovery%2Bfrom%2BCOVID-19%2Bin%2Bmultiple%2Bsclerosis%3A%2Ba%2Bprospective%2Band%2Blongitudinal%2Bcohort%2Bstudy%2Bof%2Bthe%2Bunited%2Bkingdom%2Bmultiple%2Bsclerosis%2Bregister&journal=Neurol%E2%80%94Neuroimmunol+Neuroinflamm.&volume=9)
- 22\.
González-HermosilloJAMartínez-LópezJPCarrillo-LampónSARuiz-OjedaDHerrera-RamírezSAmezcua-GuerraLMet al. del R Post-Acute COVID-19 Symptoms, a Potential Link with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A 6-Month Survey in a Mexican. *Cohort Brain Sci.* (2021) 11:760. 10.3390/brainsci11060760
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34201087)
- [CrossRef](https://doi.org/10.3390/brainsci11060760)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JA.%2BGonz%C3%A1lez-Hermosillo&author=JP.%2BMart%C3%ADnez-L%C3%B3pez&author=SA.%2BCarrillo-Lamp%C3%B3n&author=D.%2BRuiz-Ojeda&author=S.%2BHerrera-Ram%C3%ADrez&author=LM.%2BAmezcua-Guerra&publication_year=2021&title=del%2BR%2BPost-Acute%2BCOVID-19%2BSymptoms%2C%2Ba%2BPotential%2BLink%2Bwith%2BMyalgic%2BEncephalomyelitis%2FChronic%2BFatigue%2BSyndrome%3A%2BA%2B6-Month%2BSurvey%2Bin%2Ba%2BMexican&journal=Cohort+Brain+Sci.&volume=11)
- 23\.
GouraudCBottemanneHLahlou-LaforêtKBlanchardAGüntherSBattiSEet al. association between psychological distress, cognitive complaints, and neuropsychological status after a severe COVID-19 episode: a cross-sectional study. *Front Psychiatr.* (2021) 12:861. 10.3389/fpsyt.2021.725861
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34539470)
- [CrossRef](https://doi.org/10.3389/fpsyt.2021.725861)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BGouraud&author=H.%2BBottemanne&author=K.%2BLahlou-Lafor%C3%AAt&author=A.%2BBlanchard&author=S.%2BG%C3%BCnther&author=SE.%2BBatti&publication_year=2021&title=association%2Bbetween%2Bpsychological%2Bdistress%2C%2Bcognitive%2Bcomplaints%2C%2Band%2Bneuropsychological%2Bstatus%2Bafter%2Ba%2Bsevere%2BCOVID-19%2Bepisode%3A%2Ba%2Bcross-sectional%2Bstudy&journal=Front+Psychiatr.&volume=12)
- 24\.
GrahamELClarkJROrbanZSLimPHSzymanskiALTaylorCet al. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized COVID-19 “long haulers”. *Ann Clin Transl Neurol.* (2021) 8:1073–85. 10.1002/acn3.51350
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33755344)
- [CrossRef](https://doi.org/10.1002/acn3.51350)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=EL.%2BGraham&author=JR.%2BClark&author=ZS.%2BOrban&author=PH.%2BLim&author=AL.%2BSzymanski&author=C.%2BTaylor&publication_year=2021&title=Persistent%2Bneurologic%2Bsymptoms%2Band%2Bcognitive%2Bdysfunction%2Bin%2Bnon-hospitalized%2BCOVID-19%2B%E2%80%9Clong%2Bhaulers%E2%80%9D&journal=Ann+Clin+Transl+Neurol.&volume=8&pages=1073-85)
- 25\.
ImranJNasaPAlexanderLUpadhyaySAlanduruV. Psychological distress among survivors of moderate-to-critical COVID-19 illness: a multicentric prospective cross-sectional study. *Indian J Psychiatry.* (2021) 63:285–9. 10.4103/psychiatry.IndianJPsychiatry\_1074\_20
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34211223)
- [CrossRef](https://doi.org/10.4103/psychiatry.IndianJPsychiatry_1074_20)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BImran&author=P.%2BNasa&author=L.%2BAlexander&author=S.%2BUpadhyay&author=V.%2BAlanduru&publication_year=2021&title=Psychological%2Bdistress%2Bamong%2Bsurvivors%2Bof%2Bmoderate-to-critical%2BCOVID-19%2Billness%3A%2Ba%2Bmulticentric%2Bprospective%2Bcross-sectional%2Bstudy&journal=Indian+J+Psychiatry.&volume=63&pages=285-9)
- 26\.
LemhöferCSturmCLoudovici-KrugDBestNGutenbrunnerC. The impact of Post-COVID-syndrome on functioning—results from a community survey in patients after mild and moderate SARS-CoV-2-infections in Germany. *J Occup Med Toxicol.* (2021) 16:45. 10.1186/s12995-021-00337-9
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34620202)
- [CrossRef](https://doi.org/10.1186/s12995-021-00337-9)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BLemh%C3%B6fer&author=C.%2BSturm&author=D.%2BLoudovici-Krug&author=N.%2BBest&author=C.%2BGutenbrunner&publication_year=2021&title=The%2Bimpact%2Bof%2BPost-COVID-syndrome%2Bon%2Bfunctioning%E2%80%94results%2Bfrom%2Ba%2Bcommunity%2Bsurvey%2Bin%2Bpatients%2Bafter%2Bmild%2Band%2Bmoderate%2BSARS-CoV-2-infections%2Bin%2BGermany&journal=J+Occup+Med+Toxicol.&volume=16)
- 27\.
LombardoMDMFoppianiAPerettiGMMangiaviniLBattezzatiABertoliSet al. Long-Term coronavirus disease 2019 complications in inpatients and outpatients: a one-year follow-up cohort study. *Open Forum Infect Dis.* (2021) 8:ofab384. 10.1093/ofid/ofab384
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34386546)
- [CrossRef](https://doi.org/10.1093/ofid/ofab384)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=MDM.%2BLombardo&author=A.%2BFoppiani&author=GM.%2BPeretti&author=L.%2BMangiavini&author=A.%2BBattezzati&author=S.%2BBertoli&publication_year=2021&title=Long-Term%2Bcoronavirus%2Bdisease%2B2019%2Bcomplications%2Bin%2Binpatients%2Band%2Boutpatients%3A%2Ba%2Bone-year%2Bfollow-up%2Bcohort%2Bstudy&journal=Open+Forum+Infect+Dis.&volume=8)
- 28\.
MéndezRBalanzá-MartínezVLuperdiSCEstradaILatorreAGonzález-JiménezPet al. Short-term neuropsychiatric outcomes and quality of life in COVID-19 survivors. *J Intern Med.* (2021) 290:621–31. 10.1111/joim.13262
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33533521)
- [CrossRef](https://doi.org/10.1111/joim.13262)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BM%C3%A9ndez&author=V.%2BBalanz%C3%A1-Mart%C3%ADnez&author=SC.%2BLuperdi&author=I.%2BEstrada&author=A.%2BLatorre&author=P.%2BGonz%C3%A1lez-Jim%C3%A9nez&publication_year=2021&title=Short-term%2Bneuropsychiatric%2Boutcomes%2Band%2Bquality%2Bof%2Blife%2Bin%2BCOVID-19%2Bsurvivors&journal=J+Intern+Med.&volume=290&pages=621-31)
- 29\.
NaikSHaldarSNSonejaMMundadanNGGargPMittalAet al. Post COVID-19 sequelae: a prospective observational study from Northern India. *Drug Discov Ther.* (2021) 15:254–60. 10.5582/ddt.2021.01093
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34719599)
- [CrossRef](https://doi.org/10.5582/ddt.2021.01093)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=S.%2BNaik&author=SN.%2BHaldar&author=M.%2BSoneja&author=NG.%2BMundadan&author=P.%2BGarg&author=A.%2BMittal&publication_year=2021&title=Post%2BCOVID-19%2Bsequelae%3A%2Ba%2Bprospective%2Bobservational%2Bstudy%2Bfrom%2BNorthern%2BIndia&journal=Drug+Discov+Ther.&volume=15&pages=254-60)
- 30\.
RassVIanosiB-AZamarianLBeerRSahanicSLindnerAet al. Factors associated with impaired quality of life three months after being diagnosed with COVID-19. *Qual Life Res.* (2022) 31:1401–14. 10.1007/s11136-021-02998-9
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34580823)
- [CrossRef](https://doi.org/10.1007/s11136-021-02998-9)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=V.%2BRass&author=B-A.%2BIanosi&author=L.%2BZamarian&author=R.%2BBeer&author=S.%2BSahanic&author=A.%2BLindner&publication_year=2022&title=Factors%2Bassociated%2Bwith%2Bimpaired%2Bquality%2Bof%2Blife%2Bthree%2Bmonths%2Bafter%2Bbeing%2Bdiagnosed%2Bwith%2BCOVID-19&journal=Qual+Life+Res.&volume=31&pages=1401-14)
- 31\.
Romero-DuarteÁRivera-IzquierdoMGuerrero-Fernández de AlbaIPérez-ContrerasMFernández-MartínezNFRuiz-MonteroRet al. Sequelae, persistent symptomatology and outcomes after COVID-19 hospitalization: the ANCOHVID multicentre 6-month follow-up study. *BMC Med.* (2021) 19:129. 10.1186/s12916-021-02003-7
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34011359)
- [CrossRef](https://doi.org/10.1186/s12916-021-02003-7)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=%C3%81.%2BRomero-Duarte&author=M.%2BRivera-Izquierdo&author=I.%2BGuerrero-Fern%C3%A1ndez%2Bde%2BAlba&author=M.%2BP%C3%A9rez-Contreras&author=NF.%2BFern%C3%A1ndez-Mart%C3%ADnez&author=R.%2BRuiz-Montero&publication_year=2021&title=Sequelae%2C%2Bpersistent%2Bsymptomatology%2Band%2Boutcomes%2Bafter%2BCOVID-19%2Bhospitalization%3A%2Bthe%2BANCOHVID%2Bmulticentre%2B6-month%2Bfollow-up%2Bstudy&journal=BMC+Med.&volume=19)
- 32\.
ScherlingerMFeltenRGallaisFNazonCChatelusEPijnenburgLet al. Refining “Long-COVID” by a prospective multimodal evaluation of patients with long-term symptoms attributed to SARS-CoV-2 infection. *Infect Dis Ther.* (2021) 10:1747–63. 10.1007/s40121-021-00484-w
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34245450)
- [CrossRef](https://doi.org/10.1007/s40121-021-00484-w)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BScherlinger&author=R.%2BFelten&author=F.%2BGallais&author=C.%2BNazon&author=E.%2BChatelus&author=L.%2BPijnenburg&publication_year=2021&title=Refining%2B%E2%80%9CLong-COVID%E2%80%9D%2Bby%2Ba%2Bprospective%2Bmultimodal%2Bevaluation%2Bof%2Bpatients%2Bwith%2Blong-term%2Bsymptoms%2Battributed%2Bto%2BSARS-CoV-2%2Binfection&journal=Infect+Dis+Ther.&volume=10&pages=1747-63)
- 33\.
SimaniLRamezaniMDarazamIASagharichiMAalipourMAGhorbaniFet al. Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19. *J Neurovirol.* (2021) 27:154–9. 10.1007/s13365-021-00949-1
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33528827)
- [CrossRef](https://doi.org/10.1007/s13365-021-00949-1)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=L.%2BSimani&author=M.%2BRamezani&author=IA.%2BDarazam&author=M.%2BSagharichi&author=MA.%2BAalipour&author=F.%2BGhorbani&publication_year=2021&title=Prevalence%2Band%2Bcorrelates%2Bof%2Bchronic%2Bfatigue%2Bsyndrome%2Band%2Bpost-traumatic%2Bstress%2Bdisorder%2Bafter%2Bthe%2Boutbreak%2Bof%2Bthe%2BCOVID-19&journal=J+Neurovirol.&volume=27&pages=154-9)
- 34\.
SykesDLHoldsworthLJawadNGunasekeraPMoriceAHCrooksMG. Post-COVID-19 symptom burden: what is long-COVID and how should we manage it?*Lung.* (2021) 199:113–9. 10.1007/s00408-021-00423-z
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33569660)
- [CrossRef](https://doi.org/10.1007/s00408-021-00423-z)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=DL.%2BSykes&author=L.%2BHoldsworth&author=N.%2BJawad&author=P.%2BGunasekera&author=AH.%2BMorice&author=MG.%2BCrooks&publication_year=2021&title=Post-COVID-19%2Bsymptom%2Bburden%3A%2Bwhat%2Bis%2Blong-COVID%2Band%2Bhow%2Bshould%2Bwe%2Bmanage%2Bit%3F&journal=Lung.&volume=199&pages=113-9)
- 35\.
TaquetMDerconQLucianoSGeddesJRHusainMHarrisonPJ. Incidence, co-occurrence, and evolution of long-COVID features: A 6-month retrospective cohort study of 273,618 survivors of COVID-19. *PLoS Med.* (2021) 18:e1003773. 10.1371/journal.pmed.1003773
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34582441)
- [CrossRef](https://doi.org/10.1371/journal.pmed.1003773)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BTaquet&author=Q.%2BDercon&author=S.%2BLuciano&author=JR.%2BGeddes&author=M.%2BHusain&author=PJ.%2BHarrison&publication_year=2021&title=Incidence%2C%2Bco-occurrence%2C%2Band%2Bevolution%2Bof%2Blong-COVID%2Bfeatures%3A%2BA%2B6-month%2Bretrospective%2Bcohort%2Bstudy%2Bof%2B273%2C618%2Bsurvivors%2Bof%2BCOVID-19&journal=PLoS+Med.&volume=18)
- 36\.
TawfikHMShaabanHMTawfikAM. Post-COVID-19 syndrome in Egyptian healthcare staff: highlighting the carers sufferings. *Electron J Gen Med.* (2021) 18:em291. 10.29333/ejgm/10838
- [CrossRef](https://doi.org/10.29333/ejgm/10838)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=HM.%2BTawfik&author=HM.%2BShaaban&author=AM.%2BTawfik&publication_year=2021&title=Post-COVID-19%2Bsyndrome%2Bin%2BEgyptian%2Bhealthcare%2Bstaff%3A%2Bhighlighting%2Bthe%2Bcarers%2Bsufferings&journal=Electron+J+Gen+Med.&volume=18)
- 37\.
MorinL. The writing committee for the COMEBAC study group: four-month clinical status of a cohort of patients after hospitalization for COVID-19. *JAMA.* (2021) 325:1525–34. 10.1001/jama.2021.3331
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33729425)
- [CrossRef](https://doi.org/10.1001/jama.2021.3331)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=L.%2BMorin&publication_year=2021&title=The%2Bwriting%2Bcommittee%2Bfor%2Bthe%2BCOMEBAC%2Bstudy%2Bgroup%3A%2Bfour-month%2Bclinical%2Bstatus%2Bof%2Ba%2Bcohort%2Bof%2Bpatients%2Bafter%2Bhospitalization%2Bfor%2BCOVID-19&journal=JAMA.&volume=325&pages=1525-34)
- 38\.
van den BorstBPetersJBBrinkMSchoonYBleeker-RoversCPSchersHet al. Comprehensive health assessment 3 months after recovery from acute coronavirus disease 2019 (COVID-19). *Clin Infect Dis Off Publ Infect Dis Soc Am.* (2021) 73:e1089–98. 10.1093/cid/ciaa1750
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33220049)
- [CrossRef](https://doi.org/10.1093/cid/ciaa1750)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=B.%2Bvan%2Bden%2BBorst&author=JB.%2BPeters&author=M.%2BBrink&author=Y.%2BSchoon&author=CP.%2BBleeker-Rovers&author=H.%2BSchers&publication_year=2021&title=Comprehensive%2Bhealth%2Bassessment%2B3%2Bmonths%2Bafter%2Brecovery%2Bfrom%2Bacute%2Bcoronavirus%2Bdisease%2B2019%2B%28COVID-19%29&journal=Clin+Infect+Dis+Off+Publ+Infect+Dis+Soc+Am.&volume=73&pages=e1089-98)
- 39\.
VanichkachornGNewcombRCowlCTMuradMHBreeherLMillerSet al. Post–COVID-19 syndrome (Long Haul Syndrome): description of a multidisciplinary clinic at mayo clinic and characteristics of the initial patient cohort. *Mayo Clin Proc.* (2021) 96:1782–91. 10.1016/j.mayocp.2021.04.024
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34218857)
- [CrossRef](https://doi.org/10.1016/j.mayocp.2021.04.024)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=G.%2BVanichkachorn&author=R.%2BNewcomb&author=CT.%2BCowl&author=MH.%2BMurad&author=L.%2BBreeher&author=S.%2BMiller&publication_year=2021&title=Post%E2%80%93COVID-19%2Bsyndrome%2B%28Long%2BHaul%2BSyndrome%29%3A%2Bdescription%2Bof%2Ba%2Bmultidisciplinary%2Bclinic%2Bat%2Bmayo%2Bclinic%2Band%2Bcharacteristics%2Bof%2Bthe%2Binitial%2Bpatient%2Bcohort&journal=Mayo+Clin+Proc.&volume=96&pages=1782-91)
- 40\.
VannorsdallTDBrighamEFawzyARajuSGorgoneAPletnikovaAet al. Cognitive dysfunction, psychiatric distress, and functional decline after COVID-19. *J Acad Consult-Liaison Psychiatry.* (2022) 63:133–43. 10.1016/j.jaclp.2021.10.006
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34793996)
- [CrossRef](https://doi.org/10.1016/j.jaclp.2021.10.006)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=TD.%2BVannorsdall&author=E.%2BBrigham&author=A.%2BFawzy&author=S.%2BRaju&author=A.%2BGorgone&author=A.%2BPletnikova&publication_year=2022&title=Cognitive%2Bdysfunction%2C%2Bpsychiatric%2Bdistress%2C%2Band%2Bfunctional%2Bdecline%2Bafter%2BCOVID-19&journal=J+Acad+Consult-Liaison+Psychiatry.&volume=63&pages=133-43)
- 41\.
VassaliniPSerraRTarsitaniLKoukopoulosAEBorrazzoCAlessiFet al. Depressive symptoms among individuals hospitalized with COVID-19: three-month follow-up. *Brain Sci.* (2021) 11:1175. 10.3390/brainsci11091175
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34573196)
- [CrossRef](https://doi.org/10.3390/brainsci11091175)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=P.%2BVassalini&author=R.%2BSerra&author=L.%2BTarsitani&author=AE.%2BKoukopoulos&author=C.%2BBorrazzo&author=F.%2BAlessi&publication_year=2021&title=Depressive%2Bsymptoms%2Bamong%2Bindividuals%2Bhospitalized%2Bwith%2BCOVID-19%3A%2Bthree-month%2Bfollow-up&journal=Brain+Sci.&volume=11)
- 42\.
CDC C for DC P. *Subjective Cognitive Decline—A Public Health Issue.* (2019). Available online at: <https://www.cdc.gov/aging/data/subjective-cognitive-decline-brief.html> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2019&journal=Subjective+Cognitive+Decline%E2%80%94A+Public+Health+Issue.)
- 43\.
DattaniSRitchieHRoserM. *Mental Health. Our World Data.* (2021). Available online at: <https://ourworldindata.org/mental-health> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=S.%2BDattani&author=H.%2BRitchie&author=M.%2BRoser&publication_year=2021&journal=Mental+Health.+Our+World+Data.)
- 44\.
NIMH NI of MH. Post-Traumatic Stress Disorder (PTSD). *Natl Inst Ment Health NIMH.* (2022) Available online at: <https://www.nimh.nih.gov/health/statistics/post-traumatic-stress-disorder-ptsd> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2022&title=Post-Traumatic%2BStress%2BDisorder%2B%28PTSD%29&journal=Natl+Inst+Ment+Health+NIMH.)
- 45\.
StickleyALeinsaluMDeVylderJEInoueYKoyanagiA. Sleep problems and depression among 237 023 community-dwelling adults in 46 low- and middle-income countries. *Sci Rep.* (2019) 9:12011. 10.1038/s41598-019-48334-7
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/31427590)
- [CrossRef](https://doi.org/10.1038/s41598-019-48334-7)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=A.%2BStickley&author=M.%2BLeinsalu&author=JE.%2BDeVylder&author=Y.%2BInoue&author=A.%2BKoyanagi&publication_year=2019&title=Sleep%2Bproblems%2Band%2Bdepression%2Bamong%2B237%2B023%2Bcommunity-dwelling%2Badults%2Bin%2B46%2Blow-%2Band%2Bmiddle-income%2Bcountries&journal=Sci+Rep.&volume=9)
- 46\.
WHO WHO. *Depression.* (2022). Available online at: <https://www.who.int/news-room/fact-sheets/detail/depression> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2022&journal=Depression.)
- 47\.
HatchRYoungDBarberVGriffithsJHarrisonDAWatkinsonP. Anxiety, depression and post traumatic stress disorder after critical illness: a UK-wide prospective cohort study. *Crit Care Lond Engl.* (2018) 22:310. 10.1186/s13054-018-2223-6
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/30466485)
- [CrossRef](https://doi.org/10.1186/s13054-018-2223-6)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BHatch&author=D.%2BYoung&author=V.%2BBarber&author=J.%2BGriffiths&author=DA.%2BHarrison&author=P.%2BWatkinson&publication_year=2018&title=Anxiety%2C%2Bdepression%2Band%2Bpost%2Btraumatic%2Bstress%2Bdisorder%2Bafter%2Bcritical%2Billness%3A%2Ba%2BUK-wide%2Bprospective%2Bcohort%2Bstudy&journal=Crit+Care+Lond+Engl.&volume=22)
- 48\.
BenkeCAutenriethLKAsselmannEPané-FarréCA. Lockdown, quarantine measures, and social distancing: Associations with depression, anxiety and distress at the beginning of the COVID-19 pandemic among adults from Germany. *Psychiatry Res.* (2020) 293:113462. 10.1016/j.psychres.2020.113462
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32987222)
- [CrossRef](https://doi.org/10.1016/j.psychres.2020.113462)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BBenke&author=LK.%2BAutenrieth&author=E.%2BAsselmann&author=CA.%2BPan%C3%A9-Farr%C3%A9&publication_year=2020&title=Lockdown%2C%2Bquarantine%2Bmeasures%2C%2Band%2Bsocial%2Bdistancing%3A%2BAssociations%2Bwith%2Bdepression%2C%2Banxiety%2Band%2Bdistress%2Bat%2Bthe%2Bbeginning%2Bof%2Bthe%2BCOVID-19%2Bpandemic%2Bamong%2Badults%2Bfrom%2BGermany&journal=Psychiatry+Res.&volume=293)
- 49\.
MastroberardinoMCuoghi CostantiniRDe NovellisAMPFerrariSFilippiniCLongoFet al. “It's All COVID's Fault!”: symptoms of distress among workers in an italian general hospital during the pandemic. *Int J Environ Res Public Health.* (2022) 19:7313. 10.3390/ijerph19127313
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35742555)
- [CrossRef](https://doi.org/10.3390/ijerph19127313)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BMastroberardino&author=R.%2BCuoghi%2BCostantini&author=AMP.%2BDe%2BNovellis&author=S.%2BFerrari&author=C.%2BFilippini&author=F.%2BLongo&publication_year=2022&title=%E2%80%9CIt%27s%2BAll%2BCOVID%27s%2BFault%21%E2%80%9D%3A%2Bsymptoms%2Bof%2Bdistress%2Bamong%2Bworkers%2Bin%2Ban%2Bitalian%2Bgeneral%2Bhospital%2Bduring%2Bthe%2Bpandemic&journal=Int+J+Environ+Res+Public+Health.&volume=19)
- 50\.
VindegaardNBenrosME. COVID-19 pandemic and mental health consequences: systematic review of the current evidence. *Brain Behav Immun.* (2020) 89:531–42. 10.1016/j.bbi.2020.05.048
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32485289)
- [CrossRef](https://doi.org/10.1016/j.bbi.2020.05.048)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=N.%2BVindegaard&author=ME.%2BBenros&publication_year=2020&title=COVID-19%2Bpandemic%2Band%2Bmental%2Bhealth%2Bconsequences%3A%2Bsystematic%2Breview%2Bof%2Bthe%2Bcurrent%2Bevidence&journal=Brain+Behav+Immun.&volume=89&pages=531-42)
- 51\.
LoadesMEChatburnEHigson-SweeneyNReynoldsSShafranRBrigdenAet al. Rapid systematic review: the impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19. *J Am Acad Child Adolesc Psychiatr.* (2020) 59:1218-1239. 10.1016/j.jaac.2020.05.009
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32504808)
- [CrossRef](https://doi.org/10.1016/j.jaac.2020.05.009)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=ME.%2BLoades&author=E.%2BChatburn&author=N.%2BHigson-Sweeney&author=S.%2BReynolds&author=R.%2BShafran&author=A.%2BBrigden&publication_year=2020&title=Rapid%2Bsystematic%2Breview%3A%2Bthe%2Bimpact%2Bof%2Bsocial%2Bisolation%2Band%2Bloneliness%2Bon%2Bthe%2Bmental%2Bhealth%2Bof%2Bchildren%2Band%2Badolescents%2Bin%2Bthe%2Bcontext%2Bof%2BCOVID-19&journal=J+Am+Acad+Child+Adolesc+Psychiatr.)
- 52\.
MarchiMMagariniFMChiarenzaAGaleazziGMPalomaVGarridoRet al. Experience of discrimination during COVID-19 pandemic: the impact of public health measures and psychological distress among refugees and other migrants in Europe. *BMC Public Health.* (2022) 22:942. 10.1186/s12889-022-13370-y
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35538463)
- [CrossRef](https://doi.org/10.1186/s12889-022-13370-y)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BMarchi&author=FM.%2BMagarini&author=A.%2BChiarenza&author=GM.%2BGaleazzi&author=V.%2BPaloma&author=R.%2BGarrido&publication_year=2022&title=Experience%2Bof%2Bdiscrimination%2Bduring%2BCOVID-19%2Bpandemic%3A%2Bthe%2Bimpact%2Bof%2Bpublic%2Bhealth%2Bmeasures%2Band%2Bpsychological%2Bdistress%2Bamong%2Brefugees%2Band%2Bother%2Bmigrants%2Bin%2BEurope&journal=BMC+Public+Health.&volume=22)
- 53\.
ChoutkaJJansariVHornigMIwasakiA. Unexplained post-acute infection syndromes. *Nat Med.* (2022) 28:911–23. 10.1038/s41591-022-01810-6
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35585196)
- [CrossRef](https://doi.org/10.1038/s41591-022-01810-6)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BChoutka&author=V.%2BJansari&author=M.%2BHornig&author=A.%2BIwasaki&publication_year=2022&title=Unexplained%2Bpost-acute%2Binfection%2Bsyndromes&journal=Nat+Med.&volume=28&pages=911-23)
- 54\.
RogersJPChesneyEOliverDPollakTAMcGuirePFusar-PoliPet al. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. *Lancet Psychiatry.* (2020) 7:611–27. 10.1016/S2215-0366(20)30203-0
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32437679)
- [CrossRef](https://doi.org/10.1016/S2215-0366\(20\)30203-0)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JP.%2BRogers&author=E.%2BChesney&author=D.%2BOliver&author=TA.%2BPollak&author=P.%2BMcGuire&author=P.%2BFusar-Poli&publication_year=2020&title=Psychiatric%2Band%2Bneuropsychiatric%2Bpresentations%2Bassociated%2Bwith%2Bsevere%2Bcoronavirus%2Binfections%3A%2Ba%2Bsystematic%2Breview%2Band%2Bmeta-analysis%2Bwith%2Bcomparison%2Bto%2Bthe%2BCOVID-19%2Bpandemic&journal=Lancet+Psychiatry.&volume=7&pages=611-27)
- 55\.
BalcomEFNathAPowerC. Acute and chronic neurological disorders in COVID-19: potential mechanisms of disease. *Brain J Neurol.* (2021) 144:3576–88. 10.1093/brain/awab302
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34398188)
- [CrossRef](https://doi.org/10.1093/brain/awab302)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=EF.%2BBalcom&author=A.%2BNath&author=C.%2BPower&publication_year=2021&title=Acute%2Band%2Bchronic%2Bneurological%2Bdisorders%2Bin%2BCOVID-19%3A%2Bpotential%2Bmechanisms%2Bof%2Bdisease&journal=Brain+J+Neurol.&volume=144&pages=3576-88)
- 56\.
EvansRALeavyOCRichardsonMElneimaOMcAuleyHJCShikotraAet al. Clinical characteristics with inflammation profiling of long COVID and association with 1-year recovery following hospitalisation in the UK: a prospective observational study. *Lancet Respir Med.* (2022) 10:761–75. 10.1016/S2213-2600(22)00127-8
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35472304)
- [CrossRef](https://doi.org/10.1016/S2213-2600\(22\)00127-8)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=RA.%2BEvans&author=OC.%2BLeavy&author=M.%2BRichardson&author=O.%2BElneima&author=HJC.%2BMcAuley&author=A.%2BShikotra&publication_year=2022&title=Clinical%2Bcharacteristics%2Bwith%2Binflammation%2Bprofiling%2Bof%2Blong%2BCOVID%2Band%2Bassociation%2Bwith%2B1-year%2Brecovery%2Bfollowing%2Bhospitalisation%2Bin%2Bthe%2BUK%3A%2Ba%2Bprospective%2Bobservational%2Bstudy&journal=Lancet+Respir+Med.&volume=10&pages=761-75)
- 57\.
ProalADVanElzakkerMB. Long COVID or Post-acute Sequelae of COVID-19 (PASC): An overview of biological factors that may contribute to persistent symptoms. *Front Microbiol.* (2021) 12:698169. 10.3389/fmicb.2021.698169
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34248921)
- [CrossRef](https://doi.org/10.3389/fmicb.2021.698169)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=AD.%2BProal&author=MB.%2BVanElzakker&publication_year=2021&title=Long%2BCOVID%2Bor%2BPost-acute%2BSequelae%2Bof%2BCOVID-19%2B%28PASC%29%3A%2BAn%2Boverview%2Bof%2Bbiological%2Bfactors%2Bthat%2Bmay%2Bcontribute%2Bto%2Bpersistent%2Bsymptoms&journal=Front+Microbiol.&volume=12)
- 58\.
TheoharidesTCKempurajD. Role of SARS-CoV-2 spike-protein-induced activation of microglia and mast cells in the pathogenesis of neuro-COVID. *Cells.* (2023) 12:688. 10.3390/cells12050688
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/36899824)
- [CrossRef](https://doi.org/10.3390/cells12050688)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=TC.%2BTheoharides&author=D.%2BKempuraj&publication_year=2023&title=Role%2Bof%2BSARS-CoV-2%2Bspike-protein-induced%2Bactivation%2Bof%2Bmicroglia%2Band%2Bmast%2Bcells%2Bin%2Bthe%2Bpathogenesis%2Bof%2Bneuro-COVID&journal=Cells.&volume=12)
- 59\.
WeinstockLBBrookJBWaltersASGorisAAfrinLBMolderingsGJ. Mast cell activation symptoms are prevalent in Long-COVID. *Int J Infect Dis.* (2021) 112:217–26. 10.1016/j.ijid.2021.09.043
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34563706)
- [CrossRef](https://doi.org/10.1016/j.ijid.2021.09.043)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=LB.%2BWeinstock&author=JB.%2BBrook&author=AS.%2BWalters&author=A.%2BGoris&author=LB.%2BAfrin&author=GJ.%2BMolderings&publication_year=2021&title=Mast%2Bcell%2Bactivation%2Bsymptoms%2Bare%2Bprevalent%2Bin%2BLong-COVID&journal=Int+J+Infect+Dis.&volume=112&pages=217-26)
- 60\.
SongS-TWuM-LZhangH-JSuXWangJ-H. Mast cell activation triggered by retrovirus promotes acute viral infection. *Front Microbiol.* (2022) 13:8660. 10.3389/fmicb.2022.798660
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35197951)
- [CrossRef](https://doi.org/10.3389/fmicb.2022.798660)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=S-T.%2BSong&author=M-L.%2BWu&author=H-J.%2BZhang&author=X.%2BSu&author=J-H.%2BWang&publication_year=2022&title=Mast%2Bcell%2Bactivation%2Btriggered%2Bby%2Bretrovirus%2Bpromotes%2Bacute%2Bviral%2Binfection&journal=Front+Microbiol.&volume=13)
- 61\.
AbrahamSNJohnAL. Mast cell-orchestrated immunity to pathogens. *Nat Rev Immunol.* (2010) 10:440–52. 10.1038/nri2782
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/20498670)
- [CrossRef](https://doi.org/10.1038/nri2782)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=SN.%2BAbraham&author=AL.%2BJohn&publication_year=2010&title=Mast%2Bcell-orchestrated%2Bimmunity%2Bto%2Bpathogens&journal=Nat+Rev+Immunol.&volume=10&pages=440-52)
- 62\.
HickieIDavenportTWakefieldDVollmer-ConnaUCameronBVernonSDet al. Dubbo infection outcomes study group, Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study. *BMJ.* (2006) 333:575. 10.1136/bmj.38933.585764.AE
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/16950834)
- [CrossRef](https://doi.org/10.1136/bmj.38933.585764.AE)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=I.%2BHickie&author=T.%2BDavenport&author=D.%2BWakefield&author=U.%2BVollmer-Conna&author=B.%2BCameron&author=SD.%2BVernon&publication_year=2006&title=Dubbo%2Binfection%2Boutcomes%2Bstudy%2Bgroup%2C%2BPost-infective%2Band%2Bchronic%2Bfatigue%2Bsyndromes%2Bprecipitated%2Bby%2Bviral%2Band%2Bnon-viral%2Bpathogens%3A%2Bprospective%2Bcohort%2Bstudy&journal=BMJ.&volume=333)
- 63\.
HunskarGSRortveitGLitleskareSEideGEHanevikKLangelandNet al. Prevalence of fibromyalgia 10 years after infection with Giardia lamblia: a controlled prospective cohort study. *Scand J Pain.* (2022) 22:348–55. 10.1515/sjpain-2021-0122
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34679267)
- [CrossRef](https://doi.org/10.1515/sjpain-2021-0122)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=GS.%2BHunskar&author=G.%2BRortveit&author=S.%2BLitleskare&author=GE.%2BEide&author=K.%2BHanevik&author=N.%2BLangeland&publication_year=2022&title=Prevalence%2Bof%2Bfibromyalgia%2B10%2Byears%2Bafter%2Binfection%2Bwith%2BGiardia%2Blamblia%3A%2Ba%2Bcontrolled%2Bprospective%2Bcohort%2Bstudy&journal=Scand+J+Pain.&volume=22&pages=348-55)
- 64\.
PatelHSanderBNelderMP. Long-term sequelae of West Nile virus-related illness: a systematic review. *Lancet Infect Dis.* (2015) 15:951–9. 10.1016/S1473-3099(15)00134-6
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/26163373)
- [CrossRef](https://doi.org/10.1016/S1473-3099\(15\)00134-6)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=H.%2BPatel&author=B.%2BSander&author=MP.%2BNelder&publication_year=2015&title=Long-term%2Bsequelae%2Bof%2BWest%2BNile%2Bvirus-related%2Billness%3A%2Ba%2Bsystematic%2Breview&journal=Lancet+Infect+Dis.&volume=15&pages=951-9)
- 65\.
Rodríguez-MoralesAJCardona-OspinaJAFernanda Urbano-GarzónSSebastian Hurtado-ZapataJ. Prevalence of post-chikungunya infection chronic inflammatory arthritis: a systematic review and meta-analysis. *Arthritis Care Res.* (2016) 68:1849–58. 10.1002/acr.22900
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/27015439)
- [CrossRef](https://doi.org/10.1002/acr.22900)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=AJ.%2BRodr%C3%ADguez-Morales&author=JA.%2BCardona-Ospina&author=S.%2BFernanda%2BUrbano-Garz%C3%B3n&author=J.%2BSebastian%2BHurtado-Zapata&publication_year=2016&title=Prevalence%2Bof%2Bpost-chikungunya%2Binfection%2Bchronic%2Binflammatory%2Barthritis%3A%2Ba%2Bsystematic%2Breview%2Band%2Bmeta-analysis&journal=Arthritis+Care+Res.&volume=68&pages=1849-58)
- 66\.
The PREVAIL III. Study group: a longitudinal study of Ebola Sequelae in Liberia. *N Engl J Med.* (2019) 380:924–34. 10.1056/NEJMoa1805435
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/30855742)
- [CrossRef](https://doi.org/10.1056/NEJMoa1805435)
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2019&title=Study%2Bgroup%3A%2Ba%2Blongitudinal%2Bstudy%2Bof%2BEbola%2BSequelae%2Bin%2BLiberia&journal=N+Engl+J+Med.&volume=380&pages=924-34)
- 67\.
WhitePDThomasJMAmessJCrawfordDHGroverSAKangroHOet al. Incidence, risk and prognosis of acute and chronic fatigue syndromes and psychiatric disorders after glandular fever. *Br J Psychiatry J Ment Sci.* (1998) 173:475–81. 10.1192/bjp.173.6.475
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/9926075)
- [CrossRef](https://doi.org/10.1192/bjp.173.6.475)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=PD.%2BWhite&author=JM.%2BThomas&author=J.%2BAmess&author=DH.%2BCrawford&author=SA.%2BGrover&author=HO.%2BKangro&publication_year=1998&title=Incidence%2C%2Brisk%2Band%2Bprognosis%2Bof%2Bacute%2Band%2Bchronic%2Bfatigue%2Bsyndromes%2Band%2Bpsychiatric%2Bdisorders%2Bafter%2Bglandular%2Bfever&journal=Br+J+Psychiatry+J+Ment+Sci.&volume=173&pages=475-81)
- 68\.
MacphersonKCooperKHarbourJMahalDMillerCNairnM. Experiences of living with long COVID and of accessing healthcare services: a qualitative systematic review. *BMJ Open.* (2022) 12:e050979. 10.1136/bmjopen-2021-050979
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35017239)
- [CrossRef](https://doi.org/10.1136/bmjopen-2021-050979)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=K.%2BMacpherson&author=K.%2BCooper&author=J.%2BHarbour&author=D.%2BMahal&author=C.%2BMiller&author=M.%2BNairn&publication_year=2022&title=Experiences%2Bof%2Bliving%2Bwith%2Blong%2BCOVID%2Band%2Bof%2Baccessing%2Bhealthcare%2Bservices%3A%2Ba%2Bqualitative%2Bsystematic%2Breview&journal=BMJ+Open.&volume=12)
- 69\.
PorterNJasonLA. Mindfulness meditation interventions for long COVID: biobehavioral gene expression and neuroimmune functioning. *Neuropsychiatr Dis Treat.* (2022) 18:2599–626. 10.2147/NDT.S379653
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/36387947)
- [CrossRef](https://doi.org/10.2147/NDT.S379653)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=N.%2BPorter&author=LA.%2BJason&publication_year=2022&title=Mindfulness%2Bmeditation%2Binterventions%2Bfor%2Blong%2BCOVID%3A%2Bbiobehavioral%2Bgene%2Bexpression%2Band%2Bneuroimmune%2Bfunctioning&journal=Neuropsychiatr+Dis+Treat.&volume=18&pages=2599-626)
## Summary
Keywords
Long-COVID syndrome, COVID-19, mental health, depression, anxiety, posttraumatic stress
Citation
Marchi M, Grenzi P, Serafini V, Capoccia F, Rossi F, Marrino P, Pingani L, Galeazzi GM and Ferrari S (2023) Psychiatric symptoms in Long-COVID patients: a systematic review. *Front. Psychiatry* 14:1138389. doi: [10\.3389/fpsyt.2023.1138389](http://dx.doi.org/10.3389/fpsyt.2023.1138389)
Received
05 January 2023
Accepted
29 May 2023
Published
21 June 2023
Volume
14 - 2023
Edited by
Zonglin He, Hong Kong University of Science and Technology, Hong Kong SAR, China
Reviewed by
Theoharis Constantin Theoharides, Nova Southeastern University, United States; Renata Kochhann, Hospital Moinhos de Vento, Brazil; Māris Taube, Riga Stradinš University, Latvia; Alberto Spalice, Sapienza University of Rome, Italy
Updates

Check for updates
Copyright
© 2023 Marchi, Grenzi, Serafini, Capoccia, Rossi, Marrino, Pingani, Galeazzi and Ferrari.
This is an open-access article distributed under the terms of the [Creative Commons Attribution License (CC BY)](http://creativecommons.org/licenses/by/4.0/). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
**\***Correspondence:**** Gian Maria Galeazzi [gianmaria.galeazzi@ausl.re.it](mailto:gianmaria.galeazzi@ausl.re.it)
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
### Editor & Reviewers
Edited by
[ZHZonglin He Hong Kong University of Science and Technology, Hong Kong, SAR China](https://loop.frontiersin.org/people/1057351)
Reviewed by
[ASAlberto Spalice Department of Maternal, Infantile, and Urological Sciences, Faculty of Medicine and Dentistry, Sapienza University of Rome, Italy](https://loop.frontiersin.org/people/58742)
[RKRenata Kochhann Moinhos de Vento Hospital, Brazil](https://loop.frontiersin.org/people/300845)
[TCTheoharis Constantin Theoharides Nova Southeastern University, United States](https://loop.frontiersin.org/people/23129)
[MTMāris Taube Riga Stradiņš University, Latvia](https://loop.frontiersin.org/people/1529495)
high-impact AI
## AI playbook for researchers
Your step by step support for responsible and impactful AI use
[Explore the guide](https://www.frontiersin.org/ai-playbook/home)
[Published inFrontiers in Psychiatry Public Mental Health**3\.2** impact factor **6\.2** citescore](https://www.frontiersin.org/journals/psychiatry)
[Part of a Research Topic Psychosocial, Behavioral, and Clinical Implications for Public Mental Health During the COVID-19 Pandemic **74k** views **16** articles](https://www.frontiersin.org/research-topics/44904/psychosocial-behavioral-and-clinical-implications-for-public-mental-health-during-the-covid-19-pandemic)
Article metrics
[View details](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#metrics)
62
Citations
8,3k
Views
2,1k
Downloads
[Download PDF](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/pdf)
Download other formats
- [ReadCube](http://www.readcube.com/articles/10.3389/fpsyt.2023.1138389)
- [EPUB](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/epub)
- [XML](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/xml)
Cite article
Share article
- [PDF](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/pdf)
Download other format
- [ReadCube](http://www.readcube.com/articles/10.3389/fpsyt.2023.1138389)
- [EPUB](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/epub)
- [XML](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/xml)
- [Outline](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#outline)
- [Figures](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#figures)
- [Cite](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#cite)
- [Share](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#share)
- [Metrics](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#metrics)
- [Suppl. material](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#supplementaryMaterial)
- [Abstract](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#h1)
- [Introduction](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#s1)
- [Methods](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#s2)
- [Results](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#s3)
- [Discussion](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#s4)
- [Conclusions](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#s5)
- [Data availability statement](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#h7)
- [Author contributions](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#h8)
- [Conflict of interest](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#conf1)
- [Publisher’s note](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#h10)
- [Supplementary material](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#s9)
- [References](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#h12)
Figures and Tables
- Figure 1

[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F1)
- Figure 2

[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F2)
- Figure 3

[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F3)
- Figure 4

[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F4)
- Figure 5

[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F5)
- Figure 6

[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F6)
- Table 1
Characteristics of the included studies.
[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T1)
- Table 2
Pooled prevalence of psychiatric symptoms across the included studies and worldwide prevalence.
[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T2)
- Table 3
Prevalence of psychiatric symptoms by severity of the COVID-19 infection.
[View in article](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T3)
Copy to clipboard
Citation
Copy citation
Export citation file
- [BibTex](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/bibTex)
- [EndNote](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/endNote)
- [Reference Manager](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/reference)
- [Simple Text file](https://public-pages-files-2025.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/text)
- [Facebook](https://www.facebook.com/sharer/sharer.php?u=https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full)
- [X](https://www.twitter.com/share?url=https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full)
- [LinkedIn](https://www.linkedin.com/share?url=https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full)
- [Email](<mailto:?subject=Interesting science article: Psychiatric symptoms in Long-COVID patients: a systematic review&body=Hi!!%0D%0A%0D%0AI think you might find interesting the article "Psychiatric symptoms in Long-COVID patients: a systematic review".%0D%0A%0D%0AYou can read more about it here: https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full>)
- [WeChat]()
- Data Sheet 1.docx
- Guidelines
- [Author guidelines](https://www.frontiersin.org/guidelines/author-guidelines)
- [Services for authors](https://www.frontiersin.org/for-authors/author-services)
- [Policies and publication ethics](https://www.frontiersin.org/guidelines/policies-and-publication-ethics)
- [Editor guidelines](https://www.frontiersin.org/guidelines/editor-guidelines)
- [Fee policy](https://www.frontiersin.org/about/fee-policy)
- Explore
- [Articles](https://www.frontiersin.org/articles)
- [Research Topics](https://www.frontiersin.org/research-topics)
- [Journals](https://www.frontiersin.org/journals)
- [How we publish](https://www.frontiersin.org/about/how-we-publish)
- Outreach
- [Frontiers Forum](https://forum.frontiersin.org/)
- [Frontiers Policy Labs](https://policylabs.frontiersin.org/)
- [Frontiers for Young Minds](https://kids.frontiersin.org/)
- [Frontiers Planet Prize](https://www.frontiersin.org/about/frontiers-planet-prize)
- Connect
- [Help center](https://helpcenter.frontiersin.org/)
- [Emails and alerts](https://subscription-management.frontiersin.org/emails/preferences#block0)
- [Contact us](https://www.frontiersin.org/about/contact)
- [Submit](https://www.frontiersin.org/submission/submit)
- [Career opportunities](https://careers.frontiersin.org/)
Follow us
© 2026 Frontiers Media SA. All rights reserved.
[Privacy policy](https://www.frontiersin.org/legal/privacy-policy) \| [Terms and conditions](https://www.frontiersin.org/legal/terms-and-conditions) \| [Accessibility statement](https://www.frontiersin.org/about/accessibility-statement)
Share on WeChat
Scan with WeChat to share this article | ||||||||||||||||||
| Readable Markdown | ## Abstract
**Objective:**
People who have been infected by COVID-19 showing persistent symptoms after 4 weeks from recovery are thought to suffer from Long-COVID syndrome (LC). There is uncertainty on the clinical manifestations of LC. We undertook a systematic review to summarize the available evidence about the main psychiatric manifestations of LC.
**Method:**
PubMed (Medline), Scopus, CINHAL, PsycINFO, and EMBASE were searched until May 2022. Studies reporting estimation of emerging psychiatric symptoms and/or psychiatric diagnoses among adult people with LC were included. Pooled prevalence for each psychiatric condition was calculated in absence of control groups to compare with.
**Results:**
Thirty-three reports were included in the final selection, corresponding to 282,711 participants with LC. After 4 weeks from COVID-19 infection recovery, participants reported the following psychiatric symptoms: depression, anxiety, post-traumatic symptoms (PTS), cognitive and sleeping disturbances (i.e., insomnia or hypersomnia). The most common psychiatric manifestation resulted to be sleep disturbances, followed by depression, PTS, anxiety, and cognitive impairment (i.e., attention and memory deficits). However, some estimates were affected by important outlier effect played by one study. If study weight was not considered, the most reported condition was anxiety.
**Conclusions:**
LC may have non-specific psychiatric manifestations. More research is needed to better define LC and to differentiate it from other post-infectious or post-hospitalization syndromes.
**Systematic review registration:**
PROSPERO (CRD42022299408).
## Introduction
Long-COVID syndrome (LC) is a condition that can affect people who have recovered from Coronavirus Disease 2019 (COVID-19). This term was introduced to indicate a set of disorders that persist or occur at from 4 weeks after the elimination of the SARS-CoV-2 virus from the body ([1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B1)). The clinical features of LC are multifaceted; it has been posited that it can affect different organs and systems, causing somatic but also psychological manifestations that impact on quality of life ([2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B2)).
For most people, mild or moderate COVID-19 lasts for about 2 weeks; in some cases, though, symptoms can persist or develop after healing. Furthermore, also in people with asymptomatic infections later health problems may develop ([3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B3)–[5](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B5)).
Although progress has been made in the understanding of the clinical and epidemiological features, including the pathogenesis and complications of the acute phase of COVID-19, long-term consequences of the disease remain largely unclear ([6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6)).
Additionally, while neuropsychiatric symptoms that manifest acutely during infection, such as depression, post-traumatic symptoms \[PTS\], sleep and cognitive disturbances or anxiety, have received more attention, the medium- and long-term psychiatric outcomes in COVID-19 patients are still little known and understudied ([7](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B7), [8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8)).
In the available literature, there are highly heterogeneous research works on this topic, applying widely different sample sizes, inclusion and exclusion criteria, and duration of follow-up. In addition, patient assessment is mainly based on various assessment tools and questionnaires, self-administered in most cases, that do not provide a diagnosis of a condition with definite clinical significance.
Therefore, understanding the medium and long-term impact of COVID-19 is still far from being complete, not only in the context of a multidisciplinary approach, but even more so when focusing on specific areas such as mental health ([1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B1)).
We undertook this systematic review to summarize the available evidence about the main psychiatric manifestations of LC. A better understanding of the epidemiology of psychopathological manifestations among LC patients is crucial to develop prevention and early interventions.
## Methods
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol of this systematic review was registered with PROSPERO (CRD42022299408).
### Data sources and search strategy
We searched the PubMed (Medline), Scopus, CINHAL, PsycINFO, and EMBASE databases until May 2022, using the strategy outlined in the [Supplementary Table 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1) of the [Appendix](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1). In addition, the list of references of the included studies and of other reviews on related topics was screened to identify any other possible study deserving inclusion, and inadvertently missed during the initial literature search. No restrictions regarding language of publication or publication date were set.
### Eligibility criteria
We included experimental and observational studies reporting estimation of rates of emerging psychiatric symptoms and/or psychiatric diagnosis among adult people (i.e., ≥18 years old) with LC, without any restriction on other medical comorbidities or setting of enrolment. We excluded studies on participants already suffering from any psychiatric condition, studies assessing the presence of psychiatric symptoms before 4 weeks from COVID-19 recovery, and previous reviews, case-reports, case-series, editorial, and letters to the editor. We only included studies published in peer-reviewed journals, excluding conference abstracts and dissertations. If data from the same sample were published in multiple works, we considered only that study reporting more exhaustive information. Sample overlap was ruled out through a careful check of the registration codes as well as the place and year(s) of sampling.
Where available, outcome data from participants with other inflammatory or infectious diseases, including COVID-19 but without LC, were used as control group.
### Terms and definitions
LC was defined as either the presence or the persistence of any symptom that was not present before the infection after 4 weeks from the COVID-19 recovery. Infection from SARS-CoV-2 and recovery from the infection were defined according to the result of the real-time PCR on nasopharyngeal swab sample, or of broncho-alveolar lavage.
Psychiatric symptoms were collected from self-reporting or from validated psychometric tools. Where a psychiatric diagnosis was reported, it had to be defined according to standard operational diagnostic criteria (the Diagnostic and Statistical Manual of Mental Disorders \[DSM\] or the International Classification of Diseases \[ICD\]).
### Data collection and extraction
Four Reviewers (P.G., V.S., F.R., and F.C.) working independently preliminarily reviewed titles and abstracts of retrieved articles. The initial screening was followed by the analysis of full texts to check compliance with inclusion/exclusion criteria. All disagreements were discussed until consensus, and if consensus was not possible, another member of the team was consulted (M.M.). A standardized form was used for data extraction. Information concerning the year of publication, country, setting, characteristics of study participants (sample size, age, percentages of men and women), LC status, and the presence of psychiatric conditions in the LC groups (and, where available, in the control group) were collected by two authors (P.G. and V.S.) independently. Extraction sheets for each study were cross-checked for consistency and any disagreement was resolved by discussion within the research group.
### Statistical analyses
Where possible (i.e., there were at least two studies providing outcome data for LC and controls), quantitative data among studies were summarized using random effects meta-analysis ([9](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B9)). To summarize continuous outcome data (i.e., the scores on a psychometric tool), the pooled Hedges' g standardized mean differences (SMDs) and the corresponding 95% confidence intervals (CIs) were applied, while pooled odds ratios (ORs) and the corresponding 95% CIs ([10](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B10)) were used to report on dichotomous outcome data (i.e., presence/absence of psychiatric diagnosis or psychiatric symptoms).
If meta-analysis was not possible, we calculated the pooled prevalence of psychiatric symptoms and/or psychiatric diagnosis among LC patients. These estimates consisted in weighted-mean prevalence, raw mean prevalence, and median prevalence, with the relative lower and upper ranges across the studies included in the final selection.
The analyses were performed in R ([11](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B11)). Statistical tests were 2-sided and used a significance threshold of *p* \< 0.05.
### Risk of bias assessment and the GRADE
Bias risk in the included studies was independently assessed by three reviewers (P.G., V.S., and F.R.), using the Cochrane risk of bias tool ([12](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B12)). All disagreements were discussed until consensus, and if necessary, another member of the team was consulted (M.M.). Each item on the risk of bias assessment was scored as high, low, or unclear, and the GRADE tool was used to assess the overall certainty of evidence ([13](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B13)). Further information is available in the [Supplementary material](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1).
## Results
### Study characteristics
As shown in [Figure 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F1), from 2078 records screened on title and abstract, 114 full texts were analyzed. The review process led to the selection of 33 studies ([3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B3), [4](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B4), [6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6)–[8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8), [14](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B14)–[41](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B41)). These studies, referring to 33 different samples and involving a total of 282,711 LC participants, were included in the final selection and quantitative synthesis.
Figure 1
On an average, across the studies, 48% of participants were females (range 23.1–100%). The mean age of participants was 53.2 years (range 33.7–73.2). The selected studies were conducted in 13 countries: US (*n* = 6; 18.2%); Italy (*n* = 5; 15.2%); Egypt, France, Netherlands, Spain, UK (each *n* = 3; 9.1%); India (*n* = 2; 6.1%); Austria, China, Germany, Iran, Mexico (each *n* = 1; 3.0%).
All the studies were published in the last 2 years: 2021 (*n* = 31; 93.9%); 2022 (*n* = 2; 6.1%).
With respect to the outcomes reported, only 2 studies (6.1%) provided data about psychiatric diagnosis: one study assessed depressive and anxiety disorder (GAD) through a clinical interview, the other study used retrospective screening of the electronic clinical records to investigate prevalence of anxiety and depression, and cognitive impairment, according to the ICD-10 system. The remaining studies (*n* = 31, 93.9%) used self-reporting or other psychometric tools to measure the level of: depression or anxiety (*n* = 26, 78.8%), cognitive impairment (*n* = 16, 48.5%), PTS (*n* = 13, 39.4%), and sleep disturbances (*n* = 18, 54.5%). These studies applied dichotomization into positive/negative at the psychometric assessment based on the scales' cut-off for clinical significance, and the estimated prevalence for each study was calculated as the number of participants with score above the cut-off divided to the total number of participants assessed.
Notably, none of the studies included in the final selection applied a control group without LC. Concerning the severity of COVID-19 infection, 15 studies (45.5%) included patients hospitalized due to COVID-19 infection, 5 studies (15.2%) were performed on patients who had mild infection not requiring hospitalization, and 11 studies (33.3%) included both hospitalized and other managed outpatients. Information about infection severity was missing in 2 studies (6.1%).
All studies characteristics are summarized in [Table 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T1).
Table 1
| **Author, year** | **Country** | **Study design** | **Females %** | **Mean age** | **N** | **Severity of COVID-19 infection** | **P depression (measure)** | **P Anxiety (measure)** | **P Cognitive impairment (measure)** | **P PTS (measure)** | **P Sleep disturbances (measure)** |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ahmed et al. ([14](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B14)) | Egypt | Cohort | 54% | 46\.5 | 182 | Both hospitalized and outpatients | 0\.374 (SCL-90) | 0\.619 (SCL-90) | NR | 0\.286 (PCL-5) | 0\.648 (PSQI) |
| Aly and Saber ([15](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B15)) | Egypt | Cross-sectional | 100% | 73\.2 | 115 | NR | NR | NR | 0\.252 (self-reported) | NR | 0\.243 (self-reported) |
| Aranda et al. ([16](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B16)) | Spain | Cohort | 30% | 64 | 113 | Hospitalized | 0\.301 (BDI) | 0\.487 (STAI) | NR | 0\.788 (IES-R) | 0\.292 (NR) |
| Bai et al. ([17](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B17)) | Italy | Cohort | 36% | 57 | 377 | Both hospitalized and outpatients | 0\.106 (HADS) | 0\.188 (HADS) | 0\.202 (NR) | 0\.225 (IES-R) | NR |
| Boesl et al. ([18](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B18)) | Italy | Cohort | 67% | 45\.8 | 100 | Mild | 0\.615 (BDI) | NR | 0\.306 (MoCA) | NR | 0\.337 (ESS) |
| De Graaf et al. ([3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B3)) | Netherlands | Cohort | 37% | 60\.8 | 81 | Both hospitalized and outpatients | 0\.123 (PHQ-9) | 0\.037 (GAD-7) | 0\.160 (CFQ-25) | 0\.062 (PCL-5) | NR |
| Evans et al. ([4](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B4)) | UK | Cohort | 36% | 57\.9 | 1077 | Hospitalized | 0\.262 (PHQ-9) | 0\.235 (GAD-7) | 0\.139 (NR) | 0\.117 (PCL-5) | NR |
| Frontera et al. ([19](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B19)) | US | Case-control | 35% | 68\.5 | 280 | Hospitalized | 0\.254 (NeuroQoL) | 0\.511 (NeuroQoL) | 0\.473 (MoCA) | NR | 0\.375 (NeuroQoL) |
| Ganesh et al. ([20](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B20)) | US | Cohort | 61% | 44 | 817 | Both hospitalized and outpatients | NR | NR | NR | NR | 0\.200 (PROMIS) |
| Garjani et al. ([21](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B21)) | UK | Cohort | 82% | 50 | 165 | Mild | 0\.504 (NR)† | NR | NR | NR | NR |
| Gonzàlez-Hermosillo et al. ([22](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B22)) | Mexico | Cohort | 35% | 51 | 130 | Hospitalized | 0\.354 (self-reported) | 0\.392 (self-reported) | 0\.454 (self-reported) | NR | 0\.454 (self-reported) |
| Gouraud et al. ([23](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B23)) | France | Cohort | 29% | 60 | 100 | Hospitalized | 0\.220 (HADS) | 0\.310 (HADS) | NR | NR | NR |
| Graham et al. ([24](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B24)) | US | Case-control | 66% | 43\.7 | 50 | Mild | 0\.400 (NR)† | NR | 0\.820 (PROMIS) | NR | 0\.360 (PROMIS) |
| Gramaglia et al. ([6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6)) | Italy | Cohort | 40% | 61 | 238 | Both hospitalized and outpatients | 0\.294 (MINI) | 0\.328 (MINI) | NR | 0\.429 (NR) | NR |
| Horwitz et al. ([7](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B7)) | US | Cohort | 40% | 62 | 126 | Hospitalized | NR | NR | 0\.413 (PROMIS) | NR | 0\.349 (PROMIS) |
| Huang et al. ([8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8)) | China | Cohort | 48% | 57 | 1733 | Hospitalized | 0\.227 (interview)† | NR | NR | NR | 0\.264 (interview) |
| Imran et al. ([25](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B25)) | India | Cross-sectional | 33% | 44\.5 | 103 | Hospitalized | 0\.126 (PHQ-9) | 0\.214 (GAD-7) | NR | 0\.087 (PCL-5) | NR |
| Lemhofer et al. ([26](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B26)) | Germany | Cross-sectional | 59% | 49\.8 | 365 | Mild | NR | 0\.249 (NR) | NR | NR | 0\.301 (NR) |
| Lombardo et al. ([27](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B27)) | Italy | Cohort | 54% | 53 | 303 | Both hospitalized and outpatients | NR | NR | 0\.363 (semi-structured interview) | NR | 0\.465 (semi-structured interview) |
| Mendez et al. ([28](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B28)) | Spain | Cohort | 41% | 57 | 179 | Hospitalized | 0\.268 (PHQ-9) | 0\.296 (GAD-7) | 0\.184 (NR) | 0\.251 (DTS) | NR |
| Morin et al. ([37](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B37)) | France | Cohort | 42% | 60\.9 | 478 | Hospitalized | 0\.206 (BDI) | 0\.314 (HADS) | 0\.384 (MoCA, D2-R, Q3PC) | 0\.142 (PCL-5) | 0\.536 (ISI) |
| Naik et al. ([29](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B29)) | India | Cohort | 31% | 41\.6 | 272 | Both hospitalized and outpatients | 0\.022 (interview) | 0\.029 (interview) | NR | NR | 0\.063 (interview) |
| Rass et al. ([30](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B30)) | Austria | Cohort | 39% | 55 | 90 | Both hospitalized and outpatients | 0\.121 (HADS) | 0\.222 (HADS) | NR | 0\.100 (PCL-5) | NR |
| Romero-Duarte et al. ([31](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B31)) | Spain | Cohort | 46% | 63 | 794 | Hospitalized | 0\.044 (NR) | NR | NR | NR | 0\.049 (NR) |
| Scherlinger et al. ([32](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B32)) | France | Case-control | 66% | 40 | 30 | Mild | 0\.100 (Psychological interview) | 0\.267 (Psychological interview) | NR | 0\.300 (PCL-5) | NR |
| Simani et al. ([33](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B33)) | Iran | Cohort | 33% | 54\.62 | 120 | Hospitalized | NR | NR | NR | 0\.058 (PCL-5) | NR |
| Sykes et al., ([34](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B34)) | UK | Cohort | 34% | | 134 | Hospitalized | 0\.396 (self-reported) | 0\.478 (self-reported) | 0\.097 (self-reported) | NR | 0\.351 (self-reported) |
| Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) | Netherlands | Cohort | 58% | 39\.4 | 273618 | Both hospitalized and outpatients | 0\.155 (ICD-10)† | NR | 0\.040 (ICD-10) | NR | NR |
| Tawfik ([36](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B36)) | Egypt | Cohort | 58% | 33\.7 | 120 | Both hospitalized and outpatients | NR | NR | 0\.008 (NR) | NR | 0\.042 (NR) |
| Van den Borst et al. ([38](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B38)) | Netherlands | Cohort | 40% | 59 | 124 | Both hospitalized and outpatients | 0\.117 (HADS) | 0\.100 (HADS) | NR | 0\.073 (PCL-5) | NR |
| Vanichkachorn et al. ([39](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B39)) | US | Cohort | 68% | 45\.7 | 100 | NR | NR | NR | 0\.005 (NR) | NR | 0\.003 (NR) |
| Vannorsdall et al. ([40](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B40)) | US | Cohort | 59% | 54\.5 | 82 | Hospitalized | 0\.902 (PHQ-9) | 0\.646 (GAD-7) | 0\.805 (QDRS) | 0\.25 (IES-R) | NR |
| Vassalini et al. ([41](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B41)) | Italy | Cohort | 46% | 57 | 115 | Hospitalized | 0\.148 (PHQ-9) | NR | NR | NR | NR |
Characteristics of the included studies.
P, prevalence; PTS, post-traumatic symptoms; NR, not-reported; SCL-90, symptom checklist 90; PCL-5, PTSD checklist for DSM-5; PSQI, Pittsburgh sleep quality index; BDI, Beck depression inventory; STAI, state-trait anxiety inventory; IES-R, impact of event scale—revised; HADS, hospital anxiety and depression scale; MoCA, Montreal cognitive assessment; ESS, Epworth sleepiness scale; PHQ-9, patient health questionnaire-9; GAD-7, general anxiety disorder-7; CFQ-25, cognitive failure questionnaire-25; UK, United Kingdom; US, United States of America; NeuroQoL, health-related quality of life for clinical research in neurology; PROMIS, patient-reported outcomes measurement information system; MINI, mini-international neuropsychiatric interview; DTS, Davidson trauma scale; D2-R, D2 test of attention—revised; Q3PC, Q3PC cognitive screening questionnaire; ISI, insomnia severity index; ICD-10, international classification of diseases 10th revision; QDRS, quick dementia rating scale.
†depression and anxiety aggregated prevalence.
### Prevalence of psychiatric symptoms across the studies
[Table 2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T2) summarize the pooled prevalence estimates for each psychiatric symptom across the studies included in this review, and the population prevalence worldwide. Notably, prevalence of depression, anxiety, cognitive impairment, PTS, and sleep disturbances resulted much higher among LC patients than in the general population ([42](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B42)–[46](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B46)).
Table 2
| **Outcome** | **Weighted mean P** | **Mean P (min; max)** | **Median P** | **N studies (LC participants)** | **World P** |
|---|---|---|---|---|---|
| Depression | 0\.212 | 0\.254 (0.022; 0.902) | 0\.220 | 21 (5,079) | 0\.038 |
| Anxiety | 0\.158 | 0\.313 (0.029; 0.646) | 0\.296 | 23 (28,001) | 0\.040 |
| Cognitive impairment | 0\.042 | 0\.269 (0.005; 0.820) | 0\.227 | 16 (277,268) | 0\.011 |
| PTS | 0\.192 | 0\.218 (0.058; 0.788) | 0\.130 | 13 (3,162) | 0\.036 |
| Sleeping disturbances | 0\.270 | 0\.296 (0.003; 0.648) | 0\.319 | 18 (6,212) | 0\.038 |
Pooled prevalence of psychiatric symptoms across the included studies and worldwide prevalence.
P, prevalence; min, minimum; max, maximum; LC, Long-COVID syndrome; NA, not available; PTS, post-traumatic symptoms.
### Prevalence of depression
Twenty-one studies (63.3%) provided outcome data for depression among LC patients. The weighted mean prevalence across the studies was 0.212, that is quite similar to the unweighted mean and median prevalence (0.254 \[range: 0.022–0.902\] and 0.220, respectively), consistent with not significant outlier effect played by any of the study in the pooled estimate. [Figure 2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F2) shows comparison of the depression prevalence estimates across the studies, and the weighted mean prevalence.
Figure 2
### Prevalence of anxiety
Twenty-three studies (69.7%) provided outcome data for anxiety among LC patients. The weighted mean prevalence across the studies was 0.158 and was markedly influenced by the study from Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) with a far larger sample size. Unweighted mean and median prevalence were 0.313 (range: 0.029–0.646) and 0.296, respectively. [Figure 3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F3) shows comparison of the anxiety prevalence estimates across the studies, and the weighted mean prevalence.
Figure 3
### Prevalence of cognitive impairment
Sixteen studies (48.5%) provided outcome data for cognitive impairment among LC patients. The weighted mean prevalence across the studies was 0.042 and, again, was markedly influenced by the study from Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) with the largest sample size and providing among the three lowest estimates of anxiety prevalence. Unweighted mean and median prevalence were 0.269 (range: 0.005–0.820) and 0.227, respectively. [Figure 4](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F4) shows comparison of the cognitive impairment prevalence estimates across the studies, and the weighted mean prevalence.
Figure 4
### Prevalence of PTS
Thirteen studies (39.4%) provided outcome data for PTS among LC patients. The weighted mean prevalence across the studies was 0.192. Unweighted mean and median prevalence were 0.218 (range: 0.058–0.788) and 0.130, respectively. [Figure 5](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F5) shows comparison of the PTS prevalence estimates across the studies, and the weighted mean prevalence.
Figure 5
### Prevalence of sleep disturbances
Eighteen studies (54.5%) provided outcome data for sleep disturbances among LC patients. The weighted mean prevalence across the studies was 0.270. Unweighted mean and median prevalence were 0.296 (range: 0.003–0.648) and 0.319, respectively. [Figure 6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#F6) shows comparison of the sleep disturbances prevalence estimates across the studies, and the weighted mean prevalence.
Figure 6
### Psychiatric symptoms by COVID-19 infection severity
We examined the potential association between the severity of COVID-19 infection and the occurrence of psychiatric symptoms. For this purpose, we considered hospitalization as a proxy for severe infection, and outpatient management as indicator of mild infection. As shown in [Table 3](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#T3), the comparison of the severe, mild, and both mild and severe groups in terms of average prevalence of psychiatric symptoms did not find any statistically significant difference, suggesting that the severity of the infection is not related to the development of later psychiatric symptoms. Further analysis was conducted to compare only mild and severe patients, with the severe group consisting of studies that included at least one hospitalized patient. This analysis confirmed that there were no statistically significant differences in depression, anxiety, PTS, and sleep disturbances, but found inverse association between the severity of the infection and cognitive complaints (*p* = 0.048).
Table 3
| | **Mean P (SD) mild** | **Mean P (SD) severe** | **Mean P (SD) both** | ***p*\-value** |
|---|---|---|---|---|
| N | 5 | 15 | 11 | |
| Depression | 0\.36 (0.36) | 0\.29 (0.22) | 0\.16 (0.12) | 0\.339 |
| Anxiety | 0\.36 (0.12) | 0\.37 (0.14) | 0\.21 (0.19) | 0\.089 |
| Cognitive impairment | 0\.56 (0.36) | 0\.31 (0.16) | 0\.15 (0.14) | 0\.061 |
| PTS | NA | 0\.24 (0.28) | 0\.20 (0.15) | 0\.731 |
| Sleeping disturbances | 0\.33 (0.03) | 0\.33 (0.14) | 0\.28 (0.26) | 0\.879 |
Prevalence of psychiatric symptoms by severity of the COVID-19 infection.
P, prevalence; SD, standard deviation; N, number; PTS, post-traumatic symptoms; NA, not available.
p-value are based on multiple groups analysis of variance (ANOVA).
### Risk of bias and GRADE
A detailed summary on the risk of bias in all 33 trials has been reported in the [Appendix](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1) (see [Supplementary Figures 1](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1), [2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1)), along with an assessment of the quality of the evidence (see [Supplementary Table 2](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1)). In the GRADE system, the evidence from observational studies is initially set to low, there are then criteria that can be used either to downgrade or upgrade (see further information in the [Appendix](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#SM1)). The quality of the evidence is rated very low with serious threats related to the risk of bias and inconsistency.
## Discussion
This study set out to investigate the prevalence of psychiatric symptoms among LC patients. We found that the symptoms mostly associated with LC were depression, anxiety, cognitive and sleep disturbances, and PTS. The prevalence of these symptoms among LC patients is remarkably higher than that in the general population.
However, it is necessary to underline that, among the studies included in the final selection, there was one study ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) that had a sample size accounting for around 96% of the total number of participants included in this review. That study provided estimates for anxiety and cognitive impairments and the weighted average for these outcomes falls exactly on the value of the prevalence estimated by Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) by looking at the forest plots of anxiety and cognitive impairment, it can be easily observed that only three studies for anxiety and two studies for cognitive impairment provided estimates smaller than Taquet et al. ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)) supporting its influence on the pooled prevalence. Accordingly, for these two outcomes, the raw mean resulted higher than the weighted mean, because the former is not affected by the differences in the sample size across the studies.
Nevertheless, the relatively high pooled prevalence of psychiatric symptoms among LC patients requires a better understanding. Our analyses did not find a significant association between the severity of COVID-19 infection and psychiatric symptoms, except for a potential inverse association with symptoms of cognitive impairment. These findings align with the conclusions drawn from most of the reports included in our systematic review, which examined the relationship between infection severity and psychiatric symptoms ([6](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B6), [17](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B17), [20](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B20), [23](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B23), [27](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B27), [34](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B34), [40](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B40)). Notably, only one study reported an increased risk of depression and anxiety among individuals with the most severe form of infection ([8](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B8)). However, it is important to acknowledge that the confidence in the results is limited by the comparatively small representation of patients with mild infection, leading to low statistical power.
It should be noted that research in the pre-COVID-19 era observed that survivors after intensive care (IC) are at greater risk of developing long-term mental disorders ([47](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B47)). Particularly, anxiety, depression, and PTSD would have occurred in half of this sample of UK patients discharged from IC. Psychiatric symptoms of the post-IC syndrome fall into three broad categories: physical, cognitive, and psychological deterioration. Symptoms of physical deterioration include fatigue and insomnia, while cognitive and psychiatric symptoms include anxiety, depression, memory impairment, and PTSD. Therefore, there seems to be a significant overlap in the experience of some of the LC patients analyzed in this review with post-IC syndrome.
A considerable amount of COVID-related research also focused on the effect on mental health of public health measures (such as quarantine, lock-down, social isolation and other limitations to personal freedom), finding an association with symptoms of depression, anxiety, loneliness, psychosocial distress, and persisting post-traumatic arousal ([48](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B48), [49](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B49)). Therefore, another possible explanation for the higher prevalence of psychiatric symptoms among LC patients may be more a consequence of the imposed quarantine and other restrictions in terms of anxiety, fear, anger, and other negative emotions, regardless of specific aspects of the COVID-19 infection such as neuro- or systemic inflammation. Even if the quarantine imposed by a local health authority has not been directly associated with any psychological outcomes ([50](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B50)), it was suggested that belonging to a publicly recognized COVID-19 risk group/community would be associated with increased anxiety, depressive symptoms, self-concern, fear, increased psychosocial distress, and decreased life satisfaction. In addition, loneliness and isolation have been associated with an increased risk for various mental disorders (as well as for various somatic diseases). In the context of the COVID-19 pandemic, loneliness has been found to be predictive of depressive and anxious symptoms during the lockdown measures ([51](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B51), [52](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B52)).
COVID-19 is in fact only the most recent of many other infectious diseases that have been associated with chronic sequelae after recovering from the acute phase of infection ([53](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B53)): similarly as with LC, the underlying pathophysiological and etiological mechanisms are far from being clearly understood. The review by Choutka et al. ([53](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B53)) investigated the common characteristics between LC and other chronic infectious syndromes, finding higher prevalence of the following symptoms: intolerance to physical effort, neurocognitive and sensory impairment, persistent flu-like symptoms, disturbed sleep, myalgias, and arthralgias. The greatest analogies are with the post-acute effects described in the SARS epidemic in 2002–2004 ([54](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B54)).
All these elements are probably interrelated with each other and influential in the experience of LC patients.
### Limitations
The results of this review should be interpreted considering its limitations. First, the lack of a control group made difficult to draw considerations on the risk of psychiatric symptoms among LC patients, reducing considerably generalizability and reliability of our findings. This translated also in the impossibility to meta-analyze the results of the selected studies to detail the risk of psychiatric symptoms in patients who have had COVID-19. Second, most of the included studies reported measures of psychiatric symptoms instead of assessing psychiatric diagnoses, with a risk for diagnostic overestimation: this was partially attenuated by including only studies applying validated psychometric tools or clinical interviews. Third, there were a marked outlier effect played by one study ([35](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B35)), which implemented a sample size accounting for more than 90% of the total sample size. Even though that study resulted at low risk of bias in the assessment, it may have impacted on the pooled prevalence estimate of the outcomes reported in that study. Fourth, the condition of LC has been assessed only through a temporal criterion, that was 4 weeks after recovery from the infection. The lack of a more comprehensive definition of LC may have increase the heterogeneity in the estimates. Finally, the risk of bias was rated high or unclear in many studies, with serious threats related to the inconsistency in the estimates and to the assessment of confounders.
### Implications for research and clinical practice
The overlapping of some clinical features of LC in terms of signs and symptoms with other post-infectious syndromes and with the post-IC syndrome would suggest the involvement of shared pathophysiological pathways. The perspective of identifying a unified etiological model would lead the way toward the implementation of diagnostic markers and tailored treatments ([53](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B53)). At present, however, our understanding of the underlying pathophysiological mechanisms and etiological factors is poor, though promising studies are being conducted ([55](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B55)–[57](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B57)). For example, a recent review advanced the hypothesis that perivascular inflammation serves as the critical pathogenetic factor for LC neuropsychiatric manifestations ([58](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B58)). Indeed, SARS-CoV-2 and other viruses (such as retrovirus) showed the ability to activate brain mast cells and microglia resulting in the release of inflammatory, neurotoxic, and vasoactive mediators impacting neuronal connectivity and signal transmission ([59](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B59)–[61](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B61)). Hopefully, that may also converge to a better definition of functional and psycho-somatic syndromes, such as fibromyalgia and chronic fatigue syndrome, for which the association with viral infections has been previously proposed ([62](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B62)–[67](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B67)).
More research is therefore needed, more clearly comparing different patient groups (e.g., LC patients that were admitted to ICU vs. other ICU patients with or without other infectious diseases; LC patients vs. patients remitting from other infectious diseases) and applying prospective designs, allowing causal considerations, and providing more epidemiological details. Also, qualitative studies investigating the subjective experience of people recovered from COVID-19 are being conducted ([68](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B68)): this approach may also contribute to the understanding of the psychological mechanism contributing to the onset of psychological symptoms. Such different research methods could converge on a better conceptualization and analysis of the symptoms associated with the LC syndrome, as well as supporting the construction of a better defined and unified nomenclature.
A better understanding of the LC psycho-pathophysiology is essential to provide and improve treatment. From a therapeutic point of view, in close relation both to the traumatic component of a part of the symptoms found in LC, and to the inflammatory component (initially exerted by the infection and then self-sustained), interventions aimed at reducing the inflammatory process and reducing the excessive activation of the sympathetic nervous system through a relaxation response may be useful. For example, models of intervention involving reconditioning and mindfulness may help patients suffering from LC ([69](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#B69)). Future clinical trials on LC patients may be therefore warranted.
## Conclusions
People who have recovered from COVID-19 may experience more and persistent psychiatric symptoms. These include depression, anxiety, post-traumatic distress, cognitive and sleeping disturbances. However, there is marked heterogeneity in the literature about how these symptoms are investigated and differentiated from other post-infectious or post-hospitalization conditions. More research, particularly implementing control groups and prospective follow-up, are needed to better define psychopathology related or included into the LC syndrome.
## Statements
### Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
### Author contributions
MM, PG, and VS: conceptualization and planning and interpretation of the results. PG, VS, FC, FR, and PM: acquisition. MM: analysis of the data. MM, PG, VS, FC, FR, and PM: drafting. SF, LP, and GG: critical revision of the manuscript. All authors approved the final submitted version of the manuscript.
### Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
### Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
### Supplementary material
The Supplementary Material for this article can be found online at: [https://www.frontiersin.org/articles/10.3389/fpsyt.2023.1138389/full\#supplementary-material](https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full#supplementary-material)
## References
- 1\.
AkbarialiabadHTaghrirMHAbdollahiAGhahramaniNKumarMPaydarSet al. Long COVID, a comprehensive systematic scoping review. *Infection.* (2021) 49:1163–86. 10.1007/s15010-021-01666-x
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34319569)
- [CrossRef](https://doi.org/10.1007/s15010-021-01666-x)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=H.%2BAkbarialiabad&author=MH.%2BTaghrir&author=A.%2BAbdollahi&author=N.%2BGhahramani&author=M.%2BKumar&author=S.%2BPaydar&publication_year=2021&title=Long%2BCOVID%2C%2Ba%2Bcomprehensive%2Bsystematic%2Bscoping%2Breview&journal=Infection.&volume=49&pages=1163-86)
- 2\.
MichelenMManoharanLElkheirNChengVDagensAHastieCet al. Characterising long COVID: a living systematic review. *BMJ Glob Health.* (2021) 6:e005427. 10.1136/bmjgh-2021-005427
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34580069)
- [CrossRef](https://doi.org/10.1136/bmjgh-2021-005427)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BMichelen&author=L.%2BManoharan&author=N.%2BElkheir&author=V.%2BCheng&author=A.%2BDagens&author=C.%2BHastie&publication_year=2021&title=Characterising%2Blong%2BCOVID%3A%2Ba%2Bliving%2Bsystematic%2Breview&journal=BMJ+Glob+Health.&volume=6)
- 3\.
De GraafMAAntoniMLKuileMMArbousMSDuinisveldAJFFeltkampMCWet al. Short-term outpatient follow-up of COVID-19 patients: a multidisciplinary approach. *eClinicalMedicine.* (2021) 32:25. 10.1016/j.eclinm.2021.100731
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33532720)
- [CrossRef](https://doi.org/10.1016/j.eclinm.2021.100731)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=MA.%2BDe%2BGraaf&author=ML.%2BAntoni&author=MM.%2BKuile&author=MS.%2BArbous&author=AJF.%2BDuinisveld&author=MCW.%2BFeltkamp&publication_year=2021&title=Short-term%2Boutpatient%2Bfollow-up%2Bof%2BCOVID-19%2Bpatients%3A%2Ba%2Bmultidisciplinary%2Bapproach&journal=eClinicalMedicine.&volume=32)
- 4\.
EvansRAMcAuleyHHarrisonEMShikotraASingapuriASerenoMet al. Physical, cognitive, and mental health impacts of COVID-19 after hospitalisation (PHOSP-COVID): a UK multicentre, prospective cohort study. *Lancet Respir Med.* (2021) 9:1275–87. 10.1016/S2213-2600(21)00383-0
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34627560)
- [CrossRef](https://doi.org/10.1016/S2213-2600\(21\)00383-0)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=RA.%2BEvans&author=H.%2BMcAuley&author=EM.%2BHarrison&author=A.%2BShikotra&author=A.%2BSingapuri&author=M.%2BSereno&publication_year=2021&title=Physical%2C%2Bcognitive%2C%2Band%2Bmental%2Bhealth%2Bimpacts%2Bof%2BCOVID-19%2Bafter%2Bhospitalisation%2B%28PHOSP-COVID%29%3A%2Ba%2BUK%2Bmulticentre%2C%2Bprospective%2Bcohort%2Bstudy&journal=Lancet+Respir+Med.&volume=9&pages=1275-87)
- 5\.
FioreGFerrariSCutinoAGiorginoCValeoLGaleazziGMet al. Delirium in COVID-19 and post-liver transplant patients: an observational study. *Int J Psychiatry Clin Pract.* (2022) 26:343–51. 10.1080/13651501.2022.2026403
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35061952)
- [CrossRef](https://doi.org/10.1080/13651501.2022.2026403)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=G.%2BFiore&author=S.%2BFerrari&author=A.%2BCutino&author=C.%2BGiorgino&author=L.%2BValeo&author=GM.%2BGaleazzi&publication_year=2022&title=Delirium%2Bin%2BCOVID-19%2Band%2Bpost-liver%2Btransplant%2Bpatients%3A%2Ban%2Bobservational%2Bstudy&journal=Int+J+Psychiatry+Clin+Pract.&volume=26&pages=343-51)
- 6\.
GramagliaCGambaroEBellanMBalboPEBaricichASainaghiPPet al. Mid-term psychiatric outcomes of patients recovered from COVID-19 from an italian cohort of hospitalized patients. *Front Psychiatry.* (2021) 12:667385. 10.3389/fpsyt.2021.667385
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34177656)
- [CrossRef](https://doi.org/10.3389/fpsyt.2021.667385)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BGramaglia&author=E.%2BGambaro&author=M.%2BBellan&author=PE.%2BBalbo&author=A.%2BBaricich&author=PP.%2BSainaghi&publication_year=2021&title=Mid-term%2Bpsychiatric%2Boutcomes%2Bof%2Bpatients%2Brecovered%2Bfrom%2BCOVID-19%2Bfrom%2Ban%2Bitalian%2Bcohort%2Bof%2Bhospitalized%2Bpatients&journal=Front+Psychiatry.&volume=12)
- 7\.
HorwitzLIGarryKPreteAMSharmaSMendozaFKahanTet al. Six-month outcomes in patients hospitalized with severe COVID-19. *J Gen Intern Med.* (2021) 36:3772–7. 10.1007/s11606-021-07032-9
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34355349)
- [CrossRef](https://doi.org/10.1007/s11606-021-07032-9)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=LI.%2BHorwitz&author=K.%2BGarry&author=AM.%2BPrete&author=S.%2BSharma&author=F.%2BMendoza&author=T.%2BKahan&publication_year=2021&title=Six-month%2Boutcomes%2Bin%2Bpatients%2Bhospitalized%2Bwith%2Bsevere%2BCOVID-19&journal=J+Gen+Intern+Med.&volume=36&pages=3772-7)
- 8\.
HuangCHuangLWangYLiXRenLGuXet al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. *Lancet.* (2021) 397:220–32. 10.1016/S0140-6736(20)32656-8
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/37307841)
- [CrossRef](https://doi.org/10.1016/S0140-6736\(20\)32656-8)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BHuang&author=L.%2BHuang&author=Y.%2BWang&author=X.%2BLi&author=L.%2BRen&author=X.%2BGu&publication_year=2021&title=6-month%2Bconsequences%2Bof%2BCOVID-19%2Bin%2Bpatients%2Bdischarged%2Bfrom%2Bhospital%3A%2Ba%2Bcohort%2Bstudy&journal=Lancet.&volume=397&pages=220-32)
- 9\.
DerSimonianRLairdN. Meta-analysis in clinical trials. *Control Clin Trials.* (1986) 7:177–88. 10.1016/0197-2456(86)90046-2
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/3802833)
- [CrossRef](https://doi.org/10.1016/0197-2456\(86\)90046-2)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BDerSimonian&author=N.%2BLaird&publication_year=1986&title=Meta-analysis%2Bin%2Bclinical%2Btrials&journal=Control+Clin+Trials.&volume=7&pages=177-88)
- 10\.
HigginsJThomasJChandlerJCumpstonMLiTPageM. *Cochrane Handbook for Systematic Reviews of Interventions. version 6.2. Cochrane* (2021). Available online at: [www.training.cochrane.org/handbook](http://www.training.cochrane.org/handbook). (accessed January 28, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BHiggins&author=J.%2BThomas&author=J.%2BChandler&author=M.%2BCumpston&author=T.%2BLi&author=M.%2BPage&publication_year=2021&journal=Cochrane+Handbook+for+Systematic+Reviews+of+Interventions.+version+6.2.+Cochrane)
- 11\.
RStudio Team. *RStudio: Integrated Development Environment for R.* (2021) Available online at: <http://www.rstudio.com/>
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2021&journal=RStudio%3A+Integrated+Development+Environment+for+R.)
- 12\.
HigginsJPTAltmanDGGøtzschePCJüniPMoherDOxmanADet al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. *BMJ.* (2011) 343:d5928. 10.1136/bmj.d5928
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/22008217)
- [CrossRef](https://doi.org/10.1136/bmj.d5928)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JPT.%2BHiggins&author=DG.%2BAltman&author=PC.%2BG%C3%B8tzsche&author=P.%2BJ%C3%BCni&author=D.%2BMoher&author=AD.%2BOxman&publication_year=2011&title=The%2BCochrane%2BCollaboration%27s%2Btool%2Bfor%2Bassessing%2Brisk%2Bof%2Bbias%2Bin%2Brandomised%2Btrials&journal=BMJ.&volume=343)
- 13\.
SchünemannHBrozekJGuyattGOxmanA. *GRADE Handbook for Grading Quality of Evidence Strength of Recommendations.* (2013). Available online at: <https://gdt.gradepro.org/app/handbook/handbook.html> (accessed July 6, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=H.%2BSch%C3%BCnemann&author=J.%2BBrozek&author=G.%2BGuyatt&author=A.%2BOxman&publication_year=2013&journal=GRADE+Handbook+for+Grading+Quality+of+Evidence+Strength+of+Recommendations.)
- 14\.
AhmedGKKhedrEMHamadDAMeshrefTSHashemMMAlyMM. Long term impact of Covid-19 infection on sleep and mental health: a cross-sectional study. *Psychiatry Res.* (2021) 305:114243. 10.1016/j.psychres.2021.114243
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34673325)
- [CrossRef](https://doi.org/10.1016/j.psychres.2021.114243)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=GK.%2BAhmed&author=EM.%2BKhedr&author=DA.%2BHamad&author=TS.%2BMeshref&author=MM.%2BHashem&author=MM.%2BAly&publication_year=2021&title=Long%2Bterm%2Bimpact%2Bof%2BCovid-19%2Binfection%2Bon%2Bsleep%2Band%2Bmental%2Bhealth%3A%2Ba%2Bcross-sectional%2Bstudy&journal=Psychiatry+Res.&volume=305)
- 15\.
AlyMAEGSaberHG. Long COVID and chronic fatigue syndrome: a survey of elderly female survivors in Egypt. *Int J Clin Pract.* (2021) 75:e14886. 10.1111/ijcp.14886
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34537995)
- [CrossRef](https://doi.org/10.1111/ijcp.14886)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=MAEG.%2BAly&author=HG.%2BSaber&publication_year=2021&title=Long%2BCOVID%2Band%2Bchronic%2Bfatigue%2Bsyndrome%3A%2Ba%2Bsurvey%2Bof%2Belderly%2Bfemale%2Bsurvivors%2Bin%2BEgypt&journal=Int+J+Clin+Pract.&volume=75)
- 16\.
ArandaJOriolIMartínMFeriaLVázquezNRhymanNet al. Long-term impact of COVID-19 associated acute respiratory distress syndrome. *J Infect.* (2021) 83:581–8. 10.1016/j.jinf.2021.08.018
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34400219)
- [CrossRef](https://doi.org/10.1016/j.jinf.2021.08.018)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BAranda&author=I.%2BOriol&author=M.%2BMart%C3%ADn&author=L.%2BFeria&author=N.%2BV%C3%A1zquez&author=N.%2BRhyman&publication_year=2021&title=Long-term%2Bimpact%2Bof%2BCOVID-19%2Bassociated%2Bacute%2Brespiratory%2Bdistress%2Bsyndrome&journal=J+Infect.&volume=83&pages=581-8)
- 17\.
BaiFTomasoniDFalcinellaCBarbanottiDCastoldiRMulèGet al. Female gender is associated with long COVID syndrome: a prospective cohort study. *Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis.* (2022) 28:611. 10.1016/j.cmi.2021.11.002
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34763058)
- [CrossRef](https://doi.org/10.1016/j.cmi.2021.11.002)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=F.%2BBai&author=D.%2BTomasoni&author=C.%2BFalcinella&author=D.%2BBarbanotti&author=R.%2BCastoldi&author=G.%2BMul%C3%A8&publication_year=2022&title=Female%2Bgender%2Bis%2Bassociated%2Bwith%2Blong%2BCOVID%2Bsyndrome%3A%2Ba%2Bprospective%2Bcohort%2Bstudy&journal=Clin+Microbiol+Infect+Off+Publ+Eur+Soc+Clin+Microbiol+Infect+Dis.&volume=28)
- 18\.
BoeslFAudebertHEndresMPrüssHFrankeC. A neurological outpatient clinic for patients with post-COVID-19 syndrome—a report on the clinical presentations of the first 100 patients. *Front Neurol.* (2021) 12:405. 10.3389/fneur.2021.738405
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34603189)
- [CrossRef](https://doi.org/10.3389/fneur.2021.738405)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=F.%2BBoesl&author=H.%2BAudebert&author=M.%2BEndres&author=H.%2BPr%C3%BCss&author=C.%2BFranke&publication_year=2021&title=A%2Bneurological%2Boutpatient%2Bclinic%2Bfor%2Bpatients%2Bwith%2Bpost-COVID-19%2Bsyndrome%E2%80%94a%2Breport%2Bon%2Bthe%2Bclinical%2Bpresentations%2Bof%2Bthe%2Bfirst%2B100%2Bpatients&journal=Front+Neurol.&volume=12)
- 19\.
FronteraJAYangDLewisAPatelPMedicherlaCArenaVet al. A prospective study of long-term outcomes among hospitalized COVID-19 patients with and without neurological complications. *J Neurol Sci.* (2021) 426:7486. 10.1016/j.jns.2021.117486
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34000678)
- [CrossRef](https://doi.org/10.1016/j.jns.2021.117486)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JA.%2BFrontera&author=D.%2BYang&author=A.%2BLewis&author=P.%2BPatel&author=C.%2BMedicherla&author=V.%2BArena&publication_year=2021&title=A%2Bprospective%2Bstudy%2Bof%2Blong-term%2Boutcomes%2Bamong%2Bhospitalized%2BCOVID-19%2Bpatients%2Bwith%2Band%2Bwithout%2Bneurological%2Bcomplications&journal=J+Neurol+Sci.&volume=426)
- 20\.
GaneshRGhoshAKNymanMACroghanITGrachSLAnstineCVet al. Scales for assessment of persistent post-COVID symptoms: a cross sectional study. *J Prim Care Community Health.* (2021) 12:21501327211030412. 10.1177/21501327211030413
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34231395)
- [CrossRef](https://doi.org/10.1177/21501327211030413)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BGanesh&author=AK.%2BGhosh&author=MA.%2BNyman&author=IT.%2BCroghan&author=SL.%2BGrach&author=CV.%2BAnstine&publication_year=2021&title=Scales%2Bfor%2Bassessment%2Bof%2Bpersistent%2Bpost-COVID%2Bsymptoms%3A%2Ba%2Bcross%2Bsectional%2Bstudy&journal=J+Prim+Care+Community+Health.&volume=12)
- 21\.
GarjaniAMiddletonRMNicholasREvangelouN. Recovery from COVID-19 in multiple sclerosis: a prospective and longitudinal cohort study of the united kingdom multiple sclerosis register. *Neurol—Neuroimmunol Neuroinflamm.* (2022) 9:1118. 10.1212/NXI.0000000000001118
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34848503)
- [CrossRef](https://doi.org/10.1212/NXI.0000000000001118)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=A.%2BGarjani&author=RM.%2BMiddleton&author=R.%2BNicholas&author=N.%2BEvangelou&publication_year=2022&title=Recovery%2Bfrom%2BCOVID-19%2Bin%2Bmultiple%2Bsclerosis%3A%2Ba%2Bprospective%2Band%2Blongitudinal%2Bcohort%2Bstudy%2Bof%2Bthe%2Bunited%2Bkingdom%2Bmultiple%2Bsclerosis%2Bregister&journal=Neurol%E2%80%94Neuroimmunol+Neuroinflamm.&volume=9)
- 22\.
González-HermosilloJAMartínez-LópezJPCarrillo-LampónSARuiz-OjedaDHerrera-RamírezSAmezcua-GuerraLMet al. del R Post-Acute COVID-19 Symptoms, a Potential Link with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A 6-Month Survey in a Mexican. *Cohort Brain Sci.* (2021) 11:760. 10.3390/brainsci11060760
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34201087)
- [CrossRef](https://doi.org/10.3390/brainsci11060760)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JA.%2BGonz%C3%A1lez-Hermosillo&author=JP.%2BMart%C3%ADnez-L%C3%B3pez&author=SA.%2BCarrillo-Lamp%C3%B3n&author=D.%2BRuiz-Ojeda&author=S.%2BHerrera-Ram%C3%ADrez&author=LM.%2BAmezcua-Guerra&publication_year=2021&title=del%2BR%2BPost-Acute%2BCOVID-19%2BSymptoms%2C%2Ba%2BPotential%2BLink%2Bwith%2BMyalgic%2BEncephalomyelitis%2FChronic%2BFatigue%2BSyndrome%3A%2BA%2B6-Month%2BSurvey%2Bin%2Ba%2BMexican&journal=Cohort+Brain+Sci.&volume=11)
- 23\.
GouraudCBottemanneHLahlou-LaforêtKBlanchardAGüntherSBattiSEet al. association between psychological distress, cognitive complaints, and neuropsychological status after a severe COVID-19 episode: a cross-sectional study. *Front Psychiatr.* (2021) 12:861. 10.3389/fpsyt.2021.725861
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34539470)
- [CrossRef](https://doi.org/10.3389/fpsyt.2021.725861)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BGouraud&author=H.%2BBottemanne&author=K.%2BLahlou-Lafor%C3%AAt&author=A.%2BBlanchard&author=S.%2BG%C3%BCnther&author=SE.%2BBatti&publication_year=2021&title=association%2Bbetween%2Bpsychological%2Bdistress%2C%2Bcognitive%2Bcomplaints%2C%2Band%2Bneuropsychological%2Bstatus%2Bafter%2Ba%2Bsevere%2BCOVID-19%2Bepisode%3A%2Ba%2Bcross-sectional%2Bstudy&journal=Front+Psychiatr.&volume=12)
- 24\.
GrahamELClarkJROrbanZSLimPHSzymanskiALTaylorCet al. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized COVID-19 “long haulers”. *Ann Clin Transl Neurol.* (2021) 8:1073–85. 10.1002/acn3.51350
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33755344)
- [CrossRef](https://doi.org/10.1002/acn3.51350)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=EL.%2BGraham&author=JR.%2BClark&author=ZS.%2BOrban&author=PH.%2BLim&author=AL.%2BSzymanski&author=C.%2BTaylor&publication_year=2021&title=Persistent%2Bneurologic%2Bsymptoms%2Band%2Bcognitive%2Bdysfunction%2Bin%2Bnon-hospitalized%2BCOVID-19%2B%E2%80%9Clong%2Bhaulers%E2%80%9D&journal=Ann+Clin+Transl+Neurol.&volume=8&pages=1073-85)
- 25\.
ImranJNasaPAlexanderLUpadhyaySAlanduruV. Psychological distress among survivors of moderate-to-critical COVID-19 illness: a multicentric prospective cross-sectional study. *Indian J Psychiatry.* (2021) 63:285–9. 10.4103/psychiatry.IndianJPsychiatry\_1074\_20
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34211223)
- [CrossRef](https://doi.org/10.4103/psychiatry.IndianJPsychiatry_1074_20)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BImran&author=P.%2BNasa&author=L.%2BAlexander&author=S.%2BUpadhyay&author=V.%2BAlanduru&publication_year=2021&title=Psychological%2Bdistress%2Bamong%2Bsurvivors%2Bof%2Bmoderate-to-critical%2BCOVID-19%2Billness%3A%2Ba%2Bmulticentric%2Bprospective%2Bcross-sectional%2Bstudy&journal=Indian+J+Psychiatry.&volume=63&pages=285-9)
- 26\.
LemhöferCSturmCLoudovici-KrugDBestNGutenbrunnerC. The impact of Post-COVID-syndrome on functioning—results from a community survey in patients after mild and moderate SARS-CoV-2-infections in Germany. *J Occup Med Toxicol.* (2021) 16:45. 10.1186/s12995-021-00337-9
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34620202)
- [CrossRef](https://doi.org/10.1186/s12995-021-00337-9)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BLemh%C3%B6fer&author=C.%2BSturm&author=D.%2BLoudovici-Krug&author=N.%2BBest&author=C.%2BGutenbrunner&publication_year=2021&title=The%2Bimpact%2Bof%2BPost-COVID-syndrome%2Bon%2Bfunctioning%E2%80%94results%2Bfrom%2Ba%2Bcommunity%2Bsurvey%2Bin%2Bpatients%2Bafter%2Bmild%2Band%2Bmoderate%2BSARS-CoV-2-infections%2Bin%2BGermany&journal=J+Occup+Med+Toxicol.&volume=16)
- 27\.
LombardoMDMFoppianiAPerettiGMMangiaviniLBattezzatiABertoliSet al. Long-Term coronavirus disease 2019 complications in inpatients and outpatients: a one-year follow-up cohort study. *Open Forum Infect Dis.* (2021) 8:ofab384. 10.1093/ofid/ofab384
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34386546)
- [CrossRef](https://doi.org/10.1093/ofid/ofab384)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=MDM.%2BLombardo&author=A.%2BFoppiani&author=GM.%2BPeretti&author=L.%2BMangiavini&author=A.%2BBattezzati&author=S.%2BBertoli&publication_year=2021&title=Long-Term%2Bcoronavirus%2Bdisease%2B2019%2Bcomplications%2Bin%2Binpatients%2Band%2Boutpatients%3A%2Ba%2Bone-year%2Bfollow-up%2Bcohort%2Bstudy&journal=Open+Forum+Infect+Dis.&volume=8)
- 28\.
MéndezRBalanzá-MartínezVLuperdiSCEstradaILatorreAGonzález-JiménezPet al. Short-term neuropsychiatric outcomes and quality of life in COVID-19 survivors. *J Intern Med.* (2021) 290:621–31. 10.1111/joim.13262
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33533521)
- [CrossRef](https://doi.org/10.1111/joim.13262)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BM%C3%A9ndez&author=V.%2BBalanz%C3%A1-Mart%C3%ADnez&author=SC.%2BLuperdi&author=I.%2BEstrada&author=A.%2BLatorre&author=P.%2BGonz%C3%A1lez-Jim%C3%A9nez&publication_year=2021&title=Short-term%2Bneuropsychiatric%2Boutcomes%2Band%2Bquality%2Bof%2Blife%2Bin%2BCOVID-19%2Bsurvivors&journal=J+Intern+Med.&volume=290&pages=621-31)
- 29\.
NaikSHaldarSNSonejaMMundadanNGGargPMittalAet al. Post COVID-19 sequelae: a prospective observational study from Northern India. *Drug Discov Ther.* (2021) 15:254–60. 10.5582/ddt.2021.01093
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34719599)
- [CrossRef](https://doi.org/10.5582/ddt.2021.01093)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=S.%2BNaik&author=SN.%2BHaldar&author=M.%2BSoneja&author=NG.%2BMundadan&author=P.%2BGarg&author=A.%2BMittal&publication_year=2021&title=Post%2BCOVID-19%2Bsequelae%3A%2Ba%2Bprospective%2Bobservational%2Bstudy%2Bfrom%2BNorthern%2BIndia&journal=Drug+Discov+Ther.&volume=15&pages=254-60)
- 30\.
RassVIanosiB-AZamarianLBeerRSahanicSLindnerAet al. Factors associated with impaired quality of life three months after being diagnosed with COVID-19. *Qual Life Res.* (2022) 31:1401–14. 10.1007/s11136-021-02998-9
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34580823)
- [CrossRef](https://doi.org/10.1007/s11136-021-02998-9)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=V.%2BRass&author=B-A.%2BIanosi&author=L.%2BZamarian&author=R.%2BBeer&author=S.%2BSahanic&author=A.%2BLindner&publication_year=2022&title=Factors%2Bassociated%2Bwith%2Bimpaired%2Bquality%2Bof%2Blife%2Bthree%2Bmonths%2Bafter%2Bbeing%2Bdiagnosed%2Bwith%2BCOVID-19&journal=Qual+Life+Res.&volume=31&pages=1401-14)
- 31\.
Romero-DuarteÁRivera-IzquierdoMGuerrero-Fernández de AlbaIPérez-ContrerasMFernández-MartínezNFRuiz-MonteroRet al. Sequelae, persistent symptomatology and outcomes after COVID-19 hospitalization: the ANCOHVID multicentre 6-month follow-up study. *BMC Med.* (2021) 19:129. 10.1186/s12916-021-02003-7
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34011359)
- [CrossRef](https://doi.org/10.1186/s12916-021-02003-7)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=%C3%81.%2BRomero-Duarte&author=M.%2BRivera-Izquierdo&author=I.%2BGuerrero-Fern%C3%A1ndez%2Bde%2BAlba&author=M.%2BP%C3%A9rez-Contreras&author=NF.%2BFern%C3%A1ndez-Mart%C3%ADnez&author=R.%2BRuiz-Montero&publication_year=2021&title=Sequelae%2C%2Bpersistent%2Bsymptomatology%2Band%2Boutcomes%2Bafter%2BCOVID-19%2Bhospitalization%3A%2Bthe%2BANCOHVID%2Bmulticentre%2B6-month%2Bfollow-up%2Bstudy&journal=BMC+Med.&volume=19)
- 32\.
ScherlingerMFeltenRGallaisFNazonCChatelusEPijnenburgLet al. Refining “Long-COVID” by a prospective multimodal evaluation of patients with long-term symptoms attributed to SARS-CoV-2 infection. *Infect Dis Ther.* (2021) 10:1747–63. 10.1007/s40121-021-00484-w
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34245450)
- [CrossRef](https://doi.org/10.1007/s40121-021-00484-w)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BScherlinger&author=R.%2BFelten&author=F.%2BGallais&author=C.%2BNazon&author=E.%2BChatelus&author=L.%2BPijnenburg&publication_year=2021&title=Refining%2B%E2%80%9CLong-COVID%E2%80%9D%2Bby%2Ba%2Bprospective%2Bmultimodal%2Bevaluation%2Bof%2Bpatients%2Bwith%2Blong-term%2Bsymptoms%2Battributed%2Bto%2BSARS-CoV-2%2Binfection&journal=Infect+Dis+Ther.&volume=10&pages=1747-63)
- 33\.
SimaniLRamezaniMDarazamIASagharichiMAalipourMAGhorbaniFet al. Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19. *J Neurovirol.* (2021) 27:154–9. 10.1007/s13365-021-00949-1
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33528827)
- [CrossRef](https://doi.org/10.1007/s13365-021-00949-1)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=L.%2BSimani&author=M.%2BRamezani&author=IA.%2BDarazam&author=M.%2BSagharichi&author=MA.%2BAalipour&author=F.%2BGhorbani&publication_year=2021&title=Prevalence%2Band%2Bcorrelates%2Bof%2Bchronic%2Bfatigue%2Bsyndrome%2Band%2Bpost-traumatic%2Bstress%2Bdisorder%2Bafter%2Bthe%2Boutbreak%2Bof%2Bthe%2BCOVID-19&journal=J+Neurovirol.&volume=27&pages=154-9)
- 34\.
SykesDLHoldsworthLJawadNGunasekeraPMoriceAHCrooksMG. Post-COVID-19 symptom burden: what is long-COVID and how should we manage it?*Lung.* (2021) 199:113–9. 10.1007/s00408-021-00423-z
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33569660)
- [CrossRef](https://doi.org/10.1007/s00408-021-00423-z)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=DL.%2BSykes&author=L.%2BHoldsworth&author=N.%2BJawad&author=P.%2BGunasekera&author=AH.%2BMorice&author=MG.%2BCrooks&publication_year=2021&title=Post-COVID-19%2Bsymptom%2Bburden%3A%2Bwhat%2Bis%2Blong-COVID%2Band%2Bhow%2Bshould%2Bwe%2Bmanage%2Bit%3F&journal=Lung.&volume=199&pages=113-9)
- 35\.
TaquetMDerconQLucianoSGeddesJRHusainMHarrisonPJ. Incidence, co-occurrence, and evolution of long-COVID features: A 6-month retrospective cohort study of 273,618 survivors of COVID-19. *PLoS Med.* (2021) 18:e1003773. 10.1371/journal.pmed.1003773
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34582441)
- [CrossRef](https://doi.org/10.1371/journal.pmed.1003773)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BTaquet&author=Q.%2BDercon&author=S.%2BLuciano&author=JR.%2BGeddes&author=M.%2BHusain&author=PJ.%2BHarrison&publication_year=2021&title=Incidence%2C%2Bco-occurrence%2C%2Band%2Bevolution%2Bof%2Blong-COVID%2Bfeatures%3A%2BA%2B6-month%2Bretrospective%2Bcohort%2Bstudy%2Bof%2B273%2C618%2Bsurvivors%2Bof%2BCOVID-19&journal=PLoS+Med.&volume=18)
- 36\.
TawfikHMShaabanHMTawfikAM. Post-COVID-19 syndrome in Egyptian healthcare staff: highlighting the carers sufferings. *Electron J Gen Med.* (2021) 18:em291. 10.29333/ejgm/10838
- [CrossRef](https://doi.org/10.29333/ejgm/10838)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=HM.%2BTawfik&author=HM.%2BShaaban&author=AM.%2BTawfik&publication_year=2021&title=Post-COVID-19%2Bsyndrome%2Bin%2BEgyptian%2Bhealthcare%2Bstaff%3A%2Bhighlighting%2Bthe%2Bcarers%2Bsufferings&journal=Electron+J+Gen+Med.&volume=18)
- 37\.
MorinL. The writing committee for the COMEBAC study group: four-month clinical status of a cohort of patients after hospitalization for COVID-19. *JAMA.* (2021) 325:1525–34. 10.1001/jama.2021.3331
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33729425)
- [CrossRef](https://doi.org/10.1001/jama.2021.3331)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=L.%2BMorin&publication_year=2021&title=The%2Bwriting%2Bcommittee%2Bfor%2Bthe%2BCOMEBAC%2Bstudy%2Bgroup%3A%2Bfour-month%2Bclinical%2Bstatus%2Bof%2Ba%2Bcohort%2Bof%2Bpatients%2Bafter%2Bhospitalization%2Bfor%2BCOVID-19&journal=JAMA.&volume=325&pages=1525-34)
- 38\.
van den BorstBPetersJBBrinkMSchoonYBleeker-RoversCPSchersHet al. Comprehensive health assessment 3 months after recovery from acute coronavirus disease 2019 (COVID-19). *Clin Infect Dis Off Publ Infect Dis Soc Am.* (2021) 73:e1089–98. 10.1093/cid/ciaa1750
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/33220049)
- [CrossRef](https://doi.org/10.1093/cid/ciaa1750)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=B.%2Bvan%2Bden%2BBorst&author=JB.%2BPeters&author=M.%2BBrink&author=Y.%2BSchoon&author=CP.%2BBleeker-Rovers&author=H.%2BSchers&publication_year=2021&title=Comprehensive%2Bhealth%2Bassessment%2B3%2Bmonths%2Bafter%2Brecovery%2Bfrom%2Bacute%2Bcoronavirus%2Bdisease%2B2019%2B%28COVID-19%29&journal=Clin+Infect+Dis+Off+Publ+Infect+Dis+Soc+Am.&volume=73&pages=e1089-98)
- 39\.
VanichkachornGNewcombRCowlCTMuradMHBreeherLMillerSet al. Post–COVID-19 syndrome (Long Haul Syndrome): description of a multidisciplinary clinic at mayo clinic and characteristics of the initial patient cohort. *Mayo Clin Proc.* (2021) 96:1782–91. 10.1016/j.mayocp.2021.04.024
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34218857)
- [CrossRef](https://doi.org/10.1016/j.mayocp.2021.04.024)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=G.%2BVanichkachorn&author=R.%2BNewcomb&author=CT.%2BCowl&author=MH.%2BMurad&author=L.%2BBreeher&author=S.%2BMiller&publication_year=2021&title=Post%E2%80%93COVID-19%2Bsyndrome%2B%28Long%2BHaul%2BSyndrome%29%3A%2Bdescription%2Bof%2Ba%2Bmultidisciplinary%2Bclinic%2Bat%2Bmayo%2Bclinic%2Band%2Bcharacteristics%2Bof%2Bthe%2Binitial%2Bpatient%2Bcohort&journal=Mayo+Clin+Proc.&volume=96&pages=1782-91)
- 40\.
VannorsdallTDBrighamEFawzyARajuSGorgoneAPletnikovaAet al. Cognitive dysfunction, psychiatric distress, and functional decline after COVID-19. *J Acad Consult-Liaison Psychiatry.* (2022) 63:133–43. 10.1016/j.jaclp.2021.10.006
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34793996)
- [CrossRef](https://doi.org/10.1016/j.jaclp.2021.10.006)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=TD.%2BVannorsdall&author=E.%2BBrigham&author=A.%2BFawzy&author=S.%2BRaju&author=A.%2BGorgone&author=A.%2BPletnikova&publication_year=2022&title=Cognitive%2Bdysfunction%2C%2Bpsychiatric%2Bdistress%2C%2Band%2Bfunctional%2Bdecline%2Bafter%2BCOVID-19&journal=J+Acad+Consult-Liaison+Psychiatry.&volume=63&pages=133-43)
- 41\.
VassaliniPSerraRTarsitaniLKoukopoulosAEBorrazzoCAlessiFet al. Depressive symptoms among individuals hospitalized with COVID-19: three-month follow-up. *Brain Sci.* (2021) 11:1175. 10.3390/brainsci11091175
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34573196)
- [CrossRef](https://doi.org/10.3390/brainsci11091175)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=P.%2BVassalini&author=R.%2BSerra&author=L.%2BTarsitani&author=AE.%2BKoukopoulos&author=C.%2BBorrazzo&author=F.%2BAlessi&publication_year=2021&title=Depressive%2Bsymptoms%2Bamong%2Bindividuals%2Bhospitalized%2Bwith%2BCOVID-19%3A%2Bthree-month%2Bfollow-up&journal=Brain+Sci.&volume=11)
- 42\.
CDC C for DC P. *Subjective Cognitive Decline—A Public Health Issue.* (2019). Available online at: <https://www.cdc.gov/aging/data/subjective-cognitive-decline-brief.html> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2019&journal=Subjective+Cognitive+Decline%E2%80%94A+Public+Health+Issue.)
- 43\.
DattaniSRitchieHRoserM. *Mental Health. Our World Data.* (2021). Available online at: <https://ourworldindata.org/mental-health> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=S.%2BDattani&author=H.%2BRitchie&author=M.%2BRoser&publication_year=2021&journal=Mental+Health.+Our+World+Data.)
- 44\.
NIMH NI of MH. Post-Traumatic Stress Disorder (PTSD). *Natl Inst Ment Health NIMH.* (2022) Available online at: <https://www.nimh.nih.gov/health/statistics/post-traumatic-stress-disorder-ptsd> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2022&title=Post-Traumatic%2BStress%2BDisorder%2B%28PTSD%29&journal=Natl+Inst+Ment+Health+NIMH.)
- 45\.
StickleyALeinsaluMDeVylderJEInoueYKoyanagiA. Sleep problems and depression among 237 023 community-dwelling adults in 46 low- and middle-income countries. *Sci Rep.* (2019) 9:12011. 10.1038/s41598-019-48334-7
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/31427590)
- [CrossRef](https://doi.org/10.1038/s41598-019-48334-7)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=A.%2BStickley&author=M.%2BLeinsalu&author=JE.%2BDeVylder&author=Y.%2BInoue&author=A.%2BKoyanagi&publication_year=2019&title=Sleep%2Bproblems%2Band%2Bdepression%2Bamong%2B237%2B023%2Bcommunity-dwelling%2Badults%2Bin%2B46%2Blow-%2Band%2Bmiddle-income%2Bcountries&journal=Sci+Rep.&volume=9)
- 46\.
WHO WHO. *Depression.* (2022). Available online at: <https://www.who.int/news-room/fact-sheets/detail/depression> (accessed December 22, 2022).
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2022&journal=Depression.)
- 47\.
HatchRYoungDBarberVGriffithsJHarrisonDAWatkinsonP. Anxiety, depression and post traumatic stress disorder after critical illness: a UK-wide prospective cohort study. *Crit Care Lond Engl.* (2018) 22:310. 10.1186/s13054-018-2223-6
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/30466485)
- [CrossRef](https://doi.org/10.1186/s13054-018-2223-6)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=R.%2BHatch&author=D.%2BYoung&author=V.%2BBarber&author=J.%2BGriffiths&author=DA.%2BHarrison&author=P.%2BWatkinson&publication_year=2018&title=Anxiety%2C%2Bdepression%2Band%2Bpost%2Btraumatic%2Bstress%2Bdisorder%2Bafter%2Bcritical%2Billness%3A%2Ba%2BUK-wide%2Bprospective%2Bcohort%2Bstudy&journal=Crit+Care+Lond+Engl.&volume=22)
- 48\.
BenkeCAutenriethLKAsselmannEPané-FarréCA. Lockdown, quarantine measures, and social distancing: Associations with depression, anxiety and distress at the beginning of the COVID-19 pandemic among adults from Germany. *Psychiatry Res.* (2020) 293:113462. 10.1016/j.psychres.2020.113462
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32987222)
- [CrossRef](https://doi.org/10.1016/j.psychres.2020.113462)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=C.%2BBenke&author=LK.%2BAutenrieth&author=E.%2BAsselmann&author=CA.%2BPan%C3%A9-Farr%C3%A9&publication_year=2020&title=Lockdown%2C%2Bquarantine%2Bmeasures%2C%2Band%2Bsocial%2Bdistancing%3A%2BAssociations%2Bwith%2Bdepression%2C%2Banxiety%2Band%2Bdistress%2Bat%2Bthe%2Bbeginning%2Bof%2Bthe%2BCOVID-19%2Bpandemic%2Bamong%2Badults%2Bfrom%2BGermany&journal=Psychiatry+Res.&volume=293)
- 49\.
MastroberardinoMCuoghi CostantiniRDe NovellisAMPFerrariSFilippiniCLongoFet al. “It's All COVID's Fault!”: symptoms of distress among workers in an italian general hospital during the pandemic. *Int J Environ Res Public Health.* (2022) 19:7313. 10.3390/ijerph19127313
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35742555)
- [CrossRef](https://doi.org/10.3390/ijerph19127313)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BMastroberardino&author=R.%2BCuoghi%2BCostantini&author=AMP.%2BDe%2BNovellis&author=S.%2BFerrari&author=C.%2BFilippini&author=F.%2BLongo&publication_year=2022&title=%E2%80%9CIt%27s%2BAll%2BCOVID%27s%2BFault%21%E2%80%9D%3A%2Bsymptoms%2Bof%2Bdistress%2Bamong%2Bworkers%2Bin%2Ban%2Bitalian%2Bgeneral%2Bhospital%2Bduring%2Bthe%2Bpandemic&journal=Int+J+Environ+Res+Public+Health.&volume=19)
- 50\.
VindegaardNBenrosME. COVID-19 pandemic and mental health consequences: systematic review of the current evidence. *Brain Behav Immun.* (2020) 89:531–42. 10.1016/j.bbi.2020.05.048
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32485289)
- [CrossRef](https://doi.org/10.1016/j.bbi.2020.05.048)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=N.%2BVindegaard&author=ME.%2BBenros&publication_year=2020&title=COVID-19%2Bpandemic%2Band%2Bmental%2Bhealth%2Bconsequences%3A%2Bsystematic%2Breview%2Bof%2Bthe%2Bcurrent%2Bevidence&journal=Brain+Behav+Immun.&volume=89&pages=531-42)
- 51\.
LoadesMEChatburnEHigson-SweeneyNReynoldsSShafranRBrigdenAet al. Rapid systematic review: the impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19. *J Am Acad Child Adolesc Psychiatr.* (2020) 59:1218-1239. 10.1016/j.jaac.2020.05.009
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32504808)
- [CrossRef](https://doi.org/10.1016/j.jaac.2020.05.009)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=ME.%2BLoades&author=E.%2BChatburn&author=N.%2BHigson-Sweeney&author=S.%2BReynolds&author=R.%2BShafran&author=A.%2BBrigden&publication_year=2020&title=Rapid%2Bsystematic%2Breview%3A%2Bthe%2Bimpact%2Bof%2Bsocial%2Bisolation%2Band%2Bloneliness%2Bon%2Bthe%2Bmental%2Bhealth%2Bof%2Bchildren%2Band%2Badolescents%2Bin%2Bthe%2Bcontext%2Bof%2BCOVID-19&journal=J+Am+Acad+Child+Adolesc+Psychiatr.)
- 52\.
MarchiMMagariniFMChiarenzaAGaleazziGMPalomaVGarridoRet al. Experience of discrimination during COVID-19 pandemic: the impact of public health measures and psychological distress among refugees and other migrants in Europe. *BMC Public Health.* (2022) 22:942. 10.1186/s12889-022-13370-y
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35538463)
- [CrossRef](https://doi.org/10.1186/s12889-022-13370-y)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=M.%2BMarchi&author=FM.%2BMagarini&author=A.%2BChiarenza&author=GM.%2BGaleazzi&author=V.%2BPaloma&author=R.%2BGarrido&publication_year=2022&title=Experience%2Bof%2Bdiscrimination%2Bduring%2BCOVID-19%2Bpandemic%3A%2Bthe%2Bimpact%2Bof%2Bpublic%2Bhealth%2Bmeasures%2Band%2Bpsychological%2Bdistress%2Bamong%2Brefugees%2Band%2Bother%2Bmigrants%2Bin%2BEurope&journal=BMC+Public+Health.&volume=22)
- 53\.
ChoutkaJJansariVHornigMIwasakiA. Unexplained post-acute infection syndromes. *Nat Med.* (2022) 28:911–23. 10.1038/s41591-022-01810-6
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35585196)
- [CrossRef](https://doi.org/10.1038/s41591-022-01810-6)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=J.%2BChoutka&author=V.%2BJansari&author=M.%2BHornig&author=A.%2BIwasaki&publication_year=2022&title=Unexplained%2Bpost-acute%2Binfection%2Bsyndromes&journal=Nat+Med.&volume=28&pages=911-23)
- 54\.
RogersJPChesneyEOliverDPollakTAMcGuirePFusar-PoliPet al. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. *Lancet Psychiatry.* (2020) 7:611–27. 10.1016/S2215-0366(20)30203-0
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/32437679)
- [CrossRef](https://doi.org/10.1016/S2215-0366\(20\)30203-0)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=JP.%2BRogers&author=E.%2BChesney&author=D.%2BOliver&author=TA.%2BPollak&author=P.%2BMcGuire&author=P.%2BFusar-Poli&publication_year=2020&title=Psychiatric%2Band%2Bneuropsychiatric%2Bpresentations%2Bassociated%2Bwith%2Bsevere%2Bcoronavirus%2Binfections%3A%2Ba%2Bsystematic%2Breview%2Band%2Bmeta-analysis%2Bwith%2Bcomparison%2Bto%2Bthe%2BCOVID-19%2Bpandemic&journal=Lancet+Psychiatry.&volume=7&pages=611-27)
- 55\.
BalcomEFNathAPowerC. Acute and chronic neurological disorders in COVID-19: potential mechanisms of disease. *Brain J Neurol.* (2021) 144:3576–88. 10.1093/brain/awab302
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34398188)
- [CrossRef](https://doi.org/10.1093/brain/awab302)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=EF.%2BBalcom&author=A.%2BNath&author=C.%2BPower&publication_year=2021&title=Acute%2Band%2Bchronic%2Bneurological%2Bdisorders%2Bin%2BCOVID-19%3A%2Bpotential%2Bmechanisms%2Bof%2Bdisease&journal=Brain+J+Neurol.&volume=144&pages=3576-88)
- 56\.
EvansRALeavyOCRichardsonMElneimaOMcAuleyHJCShikotraAet al. Clinical characteristics with inflammation profiling of long COVID and association with 1-year recovery following hospitalisation in the UK: a prospective observational study. *Lancet Respir Med.* (2022) 10:761–75. 10.1016/S2213-2600(22)00127-8
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35472304)
- [CrossRef](https://doi.org/10.1016/S2213-2600\(22\)00127-8)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=RA.%2BEvans&author=OC.%2BLeavy&author=M.%2BRichardson&author=O.%2BElneima&author=HJC.%2BMcAuley&author=A.%2BShikotra&publication_year=2022&title=Clinical%2Bcharacteristics%2Bwith%2Binflammation%2Bprofiling%2Bof%2Blong%2BCOVID%2Band%2Bassociation%2Bwith%2B1-year%2Brecovery%2Bfollowing%2Bhospitalisation%2Bin%2Bthe%2BUK%3A%2Ba%2Bprospective%2Bobservational%2Bstudy&journal=Lancet+Respir+Med.&volume=10&pages=761-75)
- 57\.
ProalADVanElzakkerMB. Long COVID or Post-acute Sequelae of COVID-19 (PASC): An overview of biological factors that may contribute to persistent symptoms. *Front Microbiol.* (2021) 12:698169. 10.3389/fmicb.2021.698169
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34248921)
- [CrossRef](https://doi.org/10.3389/fmicb.2021.698169)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=AD.%2BProal&author=MB.%2BVanElzakker&publication_year=2021&title=Long%2BCOVID%2Bor%2BPost-acute%2BSequelae%2Bof%2BCOVID-19%2B%28PASC%29%3A%2BAn%2Boverview%2Bof%2Bbiological%2Bfactors%2Bthat%2Bmay%2Bcontribute%2Bto%2Bpersistent%2Bsymptoms&journal=Front+Microbiol.&volume=12)
- 58\.
TheoharidesTCKempurajD. Role of SARS-CoV-2 spike-protein-induced activation of microglia and mast cells in the pathogenesis of neuro-COVID. *Cells.* (2023) 12:688. 10.3390/cells12050688
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/36899824)
- [CrossRef](https://doi.org/10.3390/cells12050688)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=TC.%2BTheoharides&author=D.%2BKempuraj&publication_year=2023&title=Role%2Bof%2BSARS-CoV-2%2Bspike-protein-induced%2Bactivation%2Bof%2Bmicroglia%2Band%2Bmast%2Bcells%2Bin%2Bthe%2Bpathogenesis%2Bof%2Bneuro-COVID&journal=Cells.&volume=12)
- 59\.
WeinstockLBBrookJBWaltersASGorisAAfrinLBMolderingsGJ. Mast cell activation symptoms are prevalent in Long-COVID. *Int J Infect Dis.* (2021) 112:217–26. 10.1016/j.ijid.2021.09.043
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34563706)
- [CrossRef](https://doi.org/10.1016/j.ijid.2021.09.043)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=LB.%2BWeinstock&author=JB.%2BBrook&author=AS.%2BWalters&author=A.%2BGoris&author=LB.%2BAfrin&author=GJ.%2BMolderings&publication_year=2021&title=Mast%2Bcell%2Bactivation%2Bsymptoms%2Bare%2Bprevalent%2Bin%2BLong-COVID&journal=Int+J+Infect+Dis.&volume=112&pages=217-26)
- 60\.
SongS-TWuM-LZhangH-JSuXWangJ-H. Mast cell activation triggered by retrovirus promotes acute viral infection. *Front Microbiol.* (2022) 13:8660. 10.3389/fmicb.2022.798660
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35197951)
- [CrossRef](https://doi.org/10.3389/fmicb.2022.798660)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=S-T.%2BSong&author=M-L.%2BWu&author=H-J.%2BZhang&author=X.%2BSu&author=J-H.%2BWang&publication_year=2022&title=Mast%2Bcell%2Bactivation%2Btriggered%2Bby%2Bretrovirus%2Bpromotes%2Bacute%2Bviral%2Binfection&journal=Front+Microbiol.&volume=13)
- 61\.
AbrahamSNJohnAL. Mast cell-orchestrated immunity to pathogens. *Nat Rev Immunol.* (2010) 10:440–52. 10.1038/nri2782
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/20498670)
- [CrossRef](https://doi.org/10.1038/nri2782)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=SN.%2BAbraham&author=AL.%2BJohn&publication_year=2010&title=Mast%2Bcell-orchestrated%2Bimmunity%2Bto%2Bpathogens&journal=Nat+Rev+Immunol.&volume=10&pages=440-52)
- 62\.
HickieIDavenportTWakefieldDVollmer-ConnaUCameronBVernonSDet al. Dubbo infection outcomes study group, Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study. *BMJ.* (2006) 333:575. 10.1136/bmj.38933.585764.AE
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/16950834)
- [CrossRef](https://doi.org/10.1136/bmj.38933.585764.AE)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=I.%2BHickie&author=T.%2BDavenport&author=D.%2BWakefield&author=U.%2BVollmer-Conna&author=B.%2BCameron&author=SD.%2BVernon&publication_year=2006&title=Dubbo%2Binfection%2Boutcomes%2Bstudy%2Bgroup%2C%2BPost-infective%2Band%2Bchronic%2Bfatigue%2Bsyndromes%2Bprecipitated%2Bby%2Bviral%2Band%2Bnon-viral%2Bpathogens%3A%2Bprospective%2Bcohort%2Bstudy&journal=BMJ.&volume=333)
- 63\.
HunskarGSRortveitGLitleskareSEideGEHanevikKLangelandNet al. Prevalence of fibromyalgia 10 years after infection with Giardia lamblia: a controlled prospective cohort study. *Scand J Pain.* (2022) 22:348–55. 10.1515/sjpain-2021-0122
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/34679267)
- [CrossRef](https://doi.org/10.1515/sjpain-2021-0122)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=GS.%2BHunskar&author=G.%2BRortveit&author=S.%2BLitleskare&author=GE.%2BEide&author=K.%2BHanevik&author=N.%2BLangeland&publication_year=2022&title=Prevalence%2Bof%2Bfibromyalgia%2B10%2Byears%2Bafter%2Binfection%2Bwith%2BGiardia%2Blamblia%3A%2Ba%2Bcontrolled%2Bprospective%2Bcohort%2Bstudy&journal=Scand+J+Pain.&volume=22&pages=348-55)
- 64\.
PatelHSanderBNelderMP. Long-term sequelae of West Nile virus-related illness: a systematic review. *Lancet Infect Dis.* (2015) 15:951–9. 10.1016/S1473-3099(15)00134-6
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/26163373)
- [CrossRef](https://doi.org/10.1016/S1473-3099\(15\)00134-6)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=H.%2BPatel&author=B.%2BSander&author=MP.%2BNelder&publication_year=2015&title=Long-term%2Bsequelae%2Bof%2BWest%2BNile%2Bvirus-related%2Billness%3A%2Ba%2Bsystematic%2Breview&journal=Lancet+Infect+Dis.&volume=15&pages=951-9)
- 65\.
Rodríguez-MoralesAJCardona-OspinaJAFernanda Urbano-GarzónSSebastian Hurtado-ZapataJ. Prevalence of post-chikungunya infection chronic inflammatory arthritis: a systematic review and meta-analysis. *Arthritis Care Res.* (2016) 68:1849–58. 10.1002/acr.22900
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/27015439)
- [CrossRef](https://doi.org/10.1002/acr.22900)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=AJ.%2BRodr%C3%ADguez-Morales&author=JA.%2BCardona-Ospina&author=S.%2BFernanda%2BUrbano-Garz%C3%B3n&author=J.%2BSebastian%2BHurtado-Zapata&publication_year=2016&title=Prevalence%2Bof%2Bpost-chikungunya%2Binfection%2Bchronic%2Binflammatory%2Barthritis%3A%2Ba%2Bsystematic%2Breview%2Band%2Bmeta-analysis&journal=Arthritis+Care+Res.&volume=68&pages=1849-58)
- 66\.
The PREVAIL III. Study group: a longitudinal study of Ebola Sequelae in Liberia. *N Engl J Med.* (2019) 380:924–34. 10.1056/NEJMoa1805435
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/30855742)
- [CrossRef](https://doi.org/10.1056/NEJMoa1805435)
- [Google Scholar](http://scholar.google.com/scholar_lookup?publication_year=2019&title=Study%2Bgroup%3A%2Ba%2Blongitudinal%2Bstudy%2Bof%2BEbola%2BSequelae%2Bin%2BLiberia&journal=N+Engl+J+Med.&volume=380&pages=924-34)
- 67\.
WhitePDThomasJMAmessJCrawfordDHGroverSAKangroHOet al. Incidence, risk and prognosis of acute and chronic fatigue syndromes and psychiatric disorders after glandular fever. *Br J Psychiatry J Ment Sci.* (1998) 173:475–81. 10.1192/bjp.173.6.475
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/9926075)
- [CrossRef](https://doi.org/10.1192/bjp.173.6.475)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=PD.%2BWhite&author=JM.%2BThomas&author=J.%2BAmess&author=DH.%2BCrawford&author=SA.%2BGrover&author=HO.%2BKangro&publication_year=1998&title=Incidence%2C%2Brisk%2Band%2Bprognosis%2Bof%2Bacute%2Band%2Bchronic%2Bfatigue%2Bsyndromes%2Band%2Bpsychiatric%2Bdisorders%2Bafter%2Bglandular%2Bfever&journal=Br+J+Psychiatry+J+Ment+Sci.&volume=173&pages=475-81)
- 68\.
MacphersonKCooperKHarbourJMahalDMillerCNairnM. Experiences of living with long COVID and of accessing healthcare services: a qualitative systematic review. *BMJ Open.* (2022) 12:e050979. 10.1136/bmjopen-2021-050979
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/35017239)
- [CrossRef](https://doi.org/10.1136/bmjopen-2021-050979)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=K.%2BMacpherson&author=K.%2BCooper&author=J.%2BHarbour&author=D.%2BMahal&author=C.%2BMiller&author=M.%2BNairn&publication_year=2022&title=Experiences%2Bof%2Bliving%2Bwith%2Blong%2BCOVID%2Band%2Bof%2Baccessing%2Bhealthcare%2Bservices%3A%2Ba%2Bqualitative%2Bsystematic%2Breview&journal=BMJ+Open.&volume=12)
- 69\.
PorterNJasonLA. Mindfulness meditation interventions for long COVID: biobehavioral gene expression and neuroimmune functioning. *Neuropsychiatr Dis Treat.* (2022) 18:2599–626. 10.2147/NDT.S379653
- [Pubmed Abstract](https://pubmed.ncbi.nlm.nih.gov/36387947)
- [CrossRef](https://doi.org/10.2147/NDT.S379653)
- [Google Scholar](http://scholar.google.com/scholar_lookup?author=N.%2BPorter&author=LA.%2BJason&publication_year=2022&title=Mindfulness%2Bmeditation%2Binterventions%2Bfor%2Blong%2BCOVID%3A%2Bbiobehavioral%2Bgene%2Bexpression%2Band%2Bneuroimmune%2Bfunctioning&journal=Neuropsychiatr+Dis+Treat.&volume=18&pages=2599-626)
Keywords
Long-COVID syndrome, COVID-19, mental health, depression, anxiety, posttraumatic stress
Citation
Marchi M, Grenzi P, Serafini V, Capoccia F, Rossi F, Marrino P, Pingani L, Galeazzi GM and Ferrari S (2023) Psychiatric symptoms in Long-COVID patients: a systematic review. *Front. Psychiatry* 14:1138389. doi: [10\.3389/fpsyt.2023.1138389](http://dx.doi.org/10.3389/fpsyt.2023.1138389)
Received
05 January 2023
Accepted
29 May 2023
Published
21 June 2023
Volume
14 - 2023
Edited by
Zonglin He, Hong Kong University of Science and Technology, Hong Kong SAR, China
Reviewed by
Theoharis Constantin Theoharides, Nova Southeastern University, United States; Renata Kochhann, Hospital Moinhos de Vento, Brazil; Māris Taube, Riga Stradinš University, Latvia; Alberto Spalice, Sapienza University of Rome, Italy
Updates
Copyright
© 2023 Marchi, Grenzi, Serafini, Capoccia, Rossi, Marrino, Pingani, Galeazzi and Ferrari.
This is an open-access article distributed under the terms of the [Creative Commons Attribution License (CC BY)](http://creativecommons.org/licenses/by/4.0/). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
**\***Correspondence:**** Gian Maria Galeazzi [gianmaria.galeazzi@ausl.re.it](mailto:gianmaria.galeazzi@ausl.re.it)
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher. | ||||||||||||||||||
| ML Classification | |||||||||||||||||||
| ML Categories |
Raw JSON{
"/Health": 962,
"/Health/Mental_Health": 955,
"/Health/Mental_Health/Anxiety_and_Stress": 452,
"/Science": 261,
"/Science/Biological_Sciences": 257,
"/Science/Biological_Sciences/Neuroscience": 254
} | ||||||||||||||||||
| ML Page Types |
Raw JSON{
"/Article": 989,
"/Article/Study_or_Research_Findings": 987
} | ||||||||||||||||||
| ML Intent Types |
Raw JSON{
"Informational": 999
} | ||||||||||||||||||
| Content Metadata | |||||||||||||||||||
| Language | en | ||||||||||||||||||
| Author | null | ||||||||||||||||||
| Publish Time | not set | ||||||||||||||||||
| Original Publish Time | 2024-04-05 12:05:18 (2 years ago) | ||||||||||||||||||
| Republished | No | ||||||||||||||||||
| Word Count (Total) | 9,368 | ||||||||||||||||||
| Word Count (Content) | 7,825 | ||||||||||||||||||
| Links | |||||||||||||||||||
| External Links | 348 | ||||||||||||||||||
| Internal Links | 124 | ||||||||||||||||||
| Technical SEO | |||||||||||||||||||
| Meta Nofollow | No | ||||||||||||||||||
| Meta Noarchive | No | ||||||||||||||||||
| JS Rendered | Yes | ||||||||||||||||||
| Redirect Target | null | ||||||||||||||||||
| Performance | |||||||||||||||||||
| Download Time (ms) | 117 | ||||||||||||||||||
| TTFB (ms) | 102 | ||||||||||||||||||
| Download Size (bytes) | 185,729 | ||||||||||||||||||
| Shard | 145 (laksa) | ||||||||||||||||||
| Root Hash | 4240109820602890345 | ||||||||||||||||||
| Unparsed URL | org,frontiersin!www,/journals/psychiatry/articles/10.3389/fpsyt.2023.1138389/full s443 | ||||||||||||||||||